What is nucleus of an atom?
The Composition of the Nucleus:
- A physicist looks at a stone table and says that it is mostly empty space inside. How is this statement true?

- Matter is made up of very small particles called atoms.
- Atoms are composed of three fundamental particles: the proton, neutron and electron.
- Figure shows the location of the protons, neutrons and electrons in an atom.
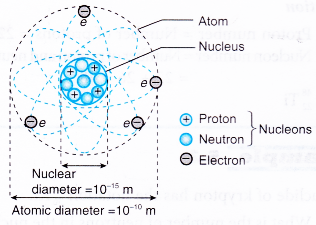
- Protons and neutrons are the nucleons. The protons and neutrons form a very small, dense core known as the nucleus. Therefore, most of the mass of an atom is contained in its nucleus.
- Electrons move in orbits around the nucleus.
- The diameter of the atom is about 100 000 times bigger than the diameter of the nucleus.
- This means that there is a lot of empty space within an atom.
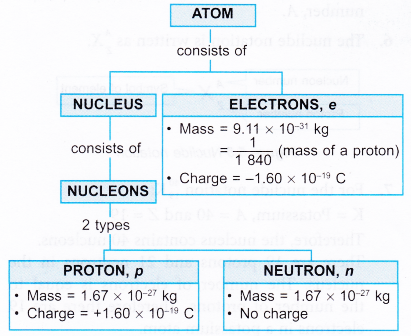
- (a) A proton is a positively-charged particle. It carries a charge of +e, where e = 1.6 × 10-19 C.
(b) A neutron carries no charge.
(c) An electron carries a negative charge of the same magnitude as the proton.
(d) Therefore, a proton and an electron are oppositely charged.
People also ask
- Radioactivity: Types of Radioactive Emissions
- How do you detect radioactivity?
- What are the different types of radioactive decay?
- What are the Isotopes, Isobars and Isotones of an Element
- What is the Radioactive Isotope?
- What is the half life of a radioactive element?
- Importance of Proper Management of Radioactive Substances
- What is Nuclear Energy?
- What is nuclear fission and how does it occur?
- How is energy released in a nuclear fusion reaction?
- What happens in a nuclear chain reaction?
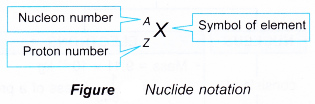
What is the symbol for nuclide?
- The proton number, Z is the number of protons in a nucleus.
- The nucleon number, A gives the total number of protons and neutrons in a nucleus.
- The number of neutrons, N = A – Z.
- A nuclide is a nucleus with a particular proton number and a particular nucleon number.
- The nuclide notation gives the symbol of the element, X, the proton number, Z and the nucleon number, A.
- The nuclide notation is written as follows:
- For the nuclide notation 19K40:
K = Potassium, A = 40 and Z = 19 Therefore, the nucleus contains 40 nucleons. There are 19 protons and 21 neutrons in the nucleus. The number of electrons is equal to the number of protons. Therefore there are 19 electrons in a potassium atom. - Table gives the number of protons, number of neutrons and the nuclide notations of some common elements.
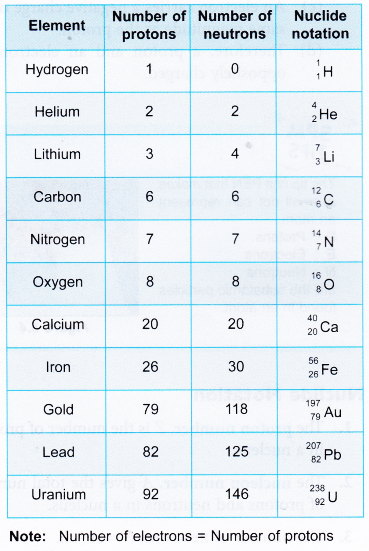
- For lighter elements such as helium, carbon and oxygen, it is common for the number of protons to be equal to the number of neutrons. Lithium is one example where the number of neutrons is not equal to the number of protons.
- Heavier elements have more neutrons than protons in the nucleus.
- The proton, neutron and electron can be represented by the following nuclide notations:

Nucleus of an Atom Examples
- A nuclide of titanium (Ti) contains 22 protons and 26 neutrons.
(a) State the proton number and nucleon number of this nuclide.
(b) Write the nuclide notation.
Solution:
(a) Proton number = Number of protons = 22
Nucleon number = Number of proton and neutrons
= 22 + 26 = 48
(b) 22Ti48 - A nuclide of krypton has the notation 36Kr84.
(a) What is the number of neutrons in the nucleus?
(b) Another nuclide of krypton has two extra neutrons. Write the nuclide notation for this nuclide.
Solution:
(a) Number of neutrons = 84 – 36 = 48
(b) Number of neutrons = 48 + 2 = 50
The proton number remains at 36 since it is the same element.
Nucleon number = 36 + 50 = 86
The nuclide notation is 36Kr84
