ICSE Solutions for Class 10 Physics – Thermionic Emission and Radioactivity
ICSE SolutionsSelina ICSE Solutions
APlusTopper.com provides ICSE Solutions for Class 10 Physics Chapter 11 Thermionic Emission and Radioactivity for ICSE Board Examinations. We provide step by step Solutions for ICSE Physics Class 10 Solutions Pdf. You can download the Class 10 Physics ICSE Textbook Solutions with Free PDF download option.
Download Formulae Handbook For ICSE Class 9 and 10
Short Answers
Question 1: Define bound electrons.
Answer: As nucleus is positively charged it strongly attracts the negative charged electrons. The electron orbit close to the nucleus are tightly bound by strong attractive force of nucleus. These electrons are known as bound electrons.
Question 2: Define free electrons.
Answer: Electrons in outer orbits are weakly bound with the nucleus. In solids these weakly bound electrons leave their individual atom and become a part of it. These electrons are known as free electrons.
Question 3: Define atomic number of an element.
Answer: The number of protons in the nucleus is known as the atomic number of the element and is denoted by Z.
Question 4: Define mass number of an element.
Answer: The total number of neutrons and protons in the nucleus is called the mass number of the element and is denoted by A.

Question 5: What are isotopes? Give examples.
Answer: The atoms of same element which have same atomic number but different mass number are called isotopes. Examples: Protium, Deuterium, Tritium.
Question 6: What are isobars? Give examples.
Answer: The atoms of different elements which have the, same mass number but differ in their atomic numbers are called isobars.
Examples: Sodium, Magnesium.
Question 7: What do you mean by electron emission?
Answer: Free electrons leave the metal surface only if sufficient energy is supplied to the metal. This emission of electrons from metal surface is called the electron emission.
Question 8: What are the types of emission?
Answer: There are four types of emissions:
(i) Thermionic emission. (ii) Photoelectric emission.
(iii) Field emission. (iv) Secondary emission.
Question 9: What do you mean by thermionic emission?
Answer: The emission of free electrons from a metal surface, when heat energy is imparted to it, is called thermionic emission.
Question 10: State three factors on which rate of emission of electrons from a metal surface depend.
Answer: (i) The material of the surface, (ii) The temperature of the surface, (iii) The surface area of the surface.
Question 11: State two characteristics of the substance used as thermionic emitter.
Answer: (i) The substance must have high melting point.
(ii) The work function of the substance should be low so that electrons may be emitted even at low temperature.
Question 12: What do you understand by the following terms?
(i) Nucleus, (ii) Nucleons.
Answer: (i) Nucleus: The central core of an atom is called nucleus. The nucleus contains protons and neutrons.
(ii) Nucleons: Neutrons and protons are collectively called nucleons.
Question 13: State two properties which a substance should possess when used as a thermionic emitter.
Answer: Two properties are:
(i) High melting point. (ii) Low work function.
Question 14: How is a cathode ray beam affected while passing through (i) a magnetic field, (ii) an electric field?
Answer: (i) A cathode ray beam while passing through a magnetic field is deflected in a direction normal to both the direction of magnetic field and the direction of its travel, as given by Fleming’s left hand rule.
(ii) A cathode ray beam while passing through an .electric field is deflected towards the positive plate (i.e. towards higher potential).
Question 15: What are cathode rays? Why are they called ‘cathode rays’? Name a material which exhibits fluorescence when cathode rays fall on it.
Answer: Cathode rays are the stream of negatively charged particles called electrons. They are called cathode rays because they are given out from cathode in a discharge tube operating at about 10,000 V when the pressure inside the tube is about 0.01 mm of Hg. Zinc Sulphide exhibits fluorescence when cathode rays fall on it.
Question 16: How is a zinc sulphide screen affected when a cathode ray beam strikes it?
Answer: When a cathode ray beam strikes a zinc sulphide screen, a bright spot is seen on the screen.
Question 17: State any two uses of cathode rays.
Answer: Cathode rays are used for:
(i) Converting electrical signal into a visual signal, (ii) In television.
Question 18: Why are materials of low work function preferred as thermionic cathode materials?
Answer: By using materials of low work functions, the emission of electrons from such a surface takes place at relatively low temperature (at 1700°C in instead of 2500°C approximately).
Question 19: In a cathode ray tube state the purpose of covering cathode by thorium and carbon.
Answer: Purpose of covering cathode by thorium and carbon is to reduce its work function, decrease its melting point.
Question 20: In a cathode ray tube state the purpose of the fluorescent screen.
Answer: Fluorescent screen helps to obtain images on the screen by converting electrical signals to light (or) visual signals by ionizing radiations to light waves.
Question 21: In a cathode ray tube state. How is it possible to increase the rate of emission of electrons.
Answer: (i) Decrease work function.
(ii) Coating the cathode with a layer of alkaline earth metallic oxides (or) hydrides.
Question 22: Name the three main parts of a Cathode Ray Tube.
Answer: The three main parts of a cathode ray tube are (i) Electron gun, (ii) Deflecting system,
(iii) Flourescent screen.
Question 23: How is a cathode ray tube used to convert an electrical signal into a visual signal?
Answer: In a CRT, the external P.D. refers to the electrical signal. The P.D. controls the deflection of electrons beam which strikes in the desired fashion direction and converts the electrical signal to visual signal.
Question 24: Name a common device where a hot cathode ray tube is used.
Answer: Hot cathode ray tube is used in T.V.
Question 25: What do you understand by the term radioactivity?
Answer: The process of self, spontaneous and random emission of a or P and y radiations from the nucleus of elements of atomic number higher than 82, is called radioactivity.
Question 26: What are ‘Becquerel rays’?
Answer: Becquerel rays are ‘radioactive radiations’, emitted from radioactive substances like uranium, thorium etc. Since these radiations were discovered by Henry Becquerel, so they are named as ‘Becquerel rays’.
Question 27: What is meant by Radioactivity?
Answer: Process of spontaneous emission of α or β and γ radiations from the nuclei of atoms during their decay.

Question 28: What are radioactive substances? Give two examples of such substances.
Answer: Radioactive substances are those which have the property of self emission of α or β and γ radiations.
Examples: Uranium, Thorium.
Question 29: Name the different radiations which are emitted by the radioactive substances.
Are all the radiations mentioned by you, emitted in a single radioactive decay?
Answer: Following three radiations are emitted by the radioactive substances:
(i) Alpha (α), (ii) Beta (β), and (iii) Gamma (γ).
All the above radiations are not emitted in a single radioactive decay. There will be either an α emission or a β emission, which may be accompained by γ-emission.

Question 30: Are all the radiations viz. α, β and γ emitted in a single radioactive decay?
Answer: All the above radiations are not emitted in a single decay. There will be either an α emission or a β emission, which may be accompanied by γ-emission.
Question 31: Compare the penetrating powers of α, β and γ-radiations.
Answer: γ -radiations are nearly 100 times more penetrating than β-radiations and 10,000 times more penetrating than α-radiations.
Question 32: Compare the ionising powers of α, β and γ radiations.
Answer: The ionising power of α-radiation is nearly 100 times that of β radiations and nearly 10,000 times that of γ-radiations.
Question 33: State the kind of nuclear reaction taking place in a nucleus during the emission of a β-particle.
Answer: In the process of emission of a β-particle from the nucleus, a neutron changes into a proton by emitting a β-particle (electron).
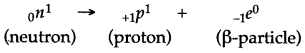
Hence in the nucleus, the number of neutrons decreases by 1 with the emission of a β-particle and the number of protons increases by one. So the sum of protons and neutrons, i.e., atomic mass remains the same but the atomic number increases by one.
Question 34: A certain nucleus has a mass number 20 and atomic number 9. Find the number of neutrons and protons present in it.
Answer: (i) Number of neutrons = Mass number – Atomic number
= 20 – 9 = 11
(ii) Number of protons = Atomic number = 9.
Question 35: Justify with reasons, whether the following nuclear reactions are allowed or not.
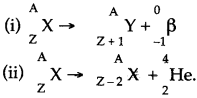
Answer: (i) This reaction is allowed because, the mass number as well as atomic number are conserved here.
(ii) This reaction is not allowed because even though charge (atomic number) is conserved in it, the mass number [= A on the L H.S. and equal to (A + 4)] on the R.H.S. is not conserved.
Question 36: An electrons emitter must have how much work function and melting point.
Answer: An electron emitter must have low work function and high melting point.
Question 37: A radioactive nucleus undergoes a series of decays according to the sequence
![]()
If the mass number and atomic number of X3 are 172 and 69 respectively, what is the mass number and atomic number of X?
Answer: Mass no. of X = 180
Atomic no. of X = 72
Question 38: A mixture of radioactive substances gives off three types of radiations:
(i) Name the radiation which travels with the speed of light.
(ii) Name the radiation which has the highest ionizing power.
Answer: (i) γ-rays.
(ii) α-particles.
Question 39: Complete the following nuclear changes:
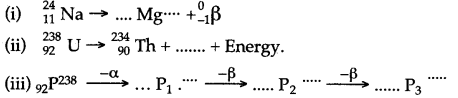
Answer:
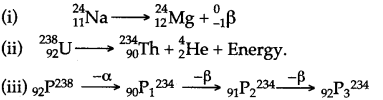
Question 40:

Answer:
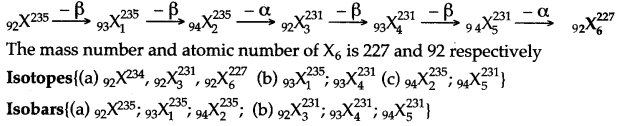
Question 41: State two factors on which the rate of emission of thermions depends.
Answer: Rate of emission of thermions depends on:
(i) Material of the surface
(ii) Temperature of the surface.
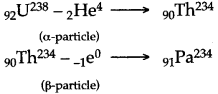
Question 42: The isotope of 92U235 decays by alpha emission to an isotope of Thorium (Th). The Thorium isotope decays by beta emission to an isotope of Protactaminum (Pa). Write down the equations to represent these two nuclear changes.
Answer:
Question 43: A mixture of radioactive substances gives off three types of radiations.
(i) Name the radiations which are charged.
(ii) Name the radiation similar in nature to cathode rays.
(iii) Name the radiation similar in nature to X-rays.
(iv) Name the radiation which is deviated most in a magnetic field.
(v) Name the radiation which is not affected by an electric field.
(vi) Name the radiation which has the maximum penetrating power.
(vii) Name the radiation which has the highest ionising power.
(viii) Name the radiation which has the highest speed.
(ix) Name the radiation which when becomes neutral, become the atom of a rare gas.
Answer: (i) Alpha and beta radiations are charged.
(ii) Beta radiation is similar in nature to cathode rays.
(iii) Gamma radiation is similar in nature to X-rays.
(iv) Beta radiation is deviated most in a magnetic field.
(v) Gamma radiation is not affected by an electric field.
(vi) Gamma radiation has the maximum penetrating power.
(vii) Alpha radiation has the highest ionising power.
(viii) Gamma radiation has the highest speed.
(ix) Alpha radiation when become neutral become the atom of rare (helium) gas.
Question 44: State, giving reasons, whether the following nuclear decays are allowed or not?
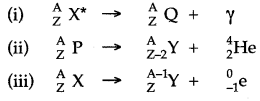
Answer: (i) This nuclear decay is allowed since the atomic number and mass number are conserved.
(ii) This nuclear decay is not allowed since the mass number is not conserved.
(iii) This nuclear decay is not allowed since the atomic number and mass number both are not conserved.

Question 46: 
![]()
Question 47: What changes will take place in the mass number and atomic number of the nucleus Y if it emits gamma radiations?
Answer: No change in mass number and atomic number when γ radiations are emitted out.
Question 48: How does the position of an element change in the periodic table when it emits out an alpha particle?
Answer: When an element emits out an alpha particle, a new element is formed with properties similar to those of an element two places earlier in the periodic table.
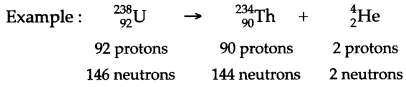
Question 49: What changes occur in the nucleus of a radioactive element when it emits an alpha particle? Show by an example?
Answer:
Question 50: What will an alpha particle change into when it absorbs:
(i) One electron; (ii) Two electrons?
Answer: (i) Single ionised Helium (He+), (ii) Helium atom.
Question 51: State three properties of α-rays.
Answer: (i) Alpha rays are composed of helium nuclei.
(ii) Alpha rays ionise the gas through which they pass.
(iii) Alpha rays are deflected by the electric and magnetic fields.
Question 52: What is the effect on the atomic mass and the atomic number of a radioactive element, after the emission of an α-particle?
Answer: During the emission of an α-particle from the nucleus of a radioactive element, its atomic mass decreases by 4 and the atomic number also decreases by 2. If ZXA represents a radioactive element having atomic mass A and atomic number Z, then:
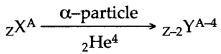
Question 53: When an alpha particle gains two electrons it becomes neutral and becomes an atom of an element which is a rare gas. What is the name of this rare gas?
Answer: Helium gas.
Question 54: An atomic nucleus A is composed of 84 protons and 128 neutrons. It emits an α-particle and is transformed into nucleus B. Write down the composition of B.
Answer: A emits an α-particle, so in the nucleus of B, neutrons will be 128 – 2 = 126 and protons will be 84 – 2 = 82.
Question 55: State charge and mass of (i) α-particle, (ii) β-particle.
Answer: (i) The charge on α-particle is 3.2 × 10-19 coulomb and its mass is 6.67 × 10-27 kg (i.e., nearly 4 times the mass of proton).
(ii) The charge on β-particle is – 1.6 × 10-19 coulomb and its mass is 9.1 × 10-31 kg.
Question 56: How does the position of an element change in the periodic table when it emits out a beta particle?
Answer: When an element emits out a beta particle, a new element is formed with properties similar to those of an element one place later in the periodic table.
Question 57: What are α-and β-radiations?
Answer: α radiations are positively charged particles with charge + 2e and mass 4mp where e is electronic charge (i.e., charge on a proton) and mp is the mass of a proton.
β radiations are negatively charged particles (i.e., electrons) with charge – e and mass 1/1836 times the mass of a proton.
γ-radiation are electromagnetic waves of wavelength in the range 0.001 Å.
Question 58: State three common properties of beta rays and cathode rays.
Answer: (i) Beta rays and cathode rays both are composed of electrons.
(ii) Beta rays and cathode rays both are deflected by the electric and magnetic fields in the same direction.
(iii) Beta rays and cathode rays both ionise the gas through which they pass.
Question 59: The nucleus of an atom does not contain electrons. Explain how is it possible for the nucleus to emit a beta particle which is fast moving electron.
Answer: The nucleus emits a beta particle when a neutron inside it changes into a proton. The reaction is
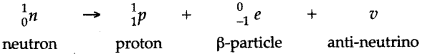
Question 60: What happens to the atomic number of an element when it emits:
(i) An alpha particle; (ii) A beta particle.
Answer:
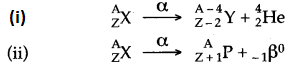
Question 61: A radioactive carbon nucleus 6C14 emits a β-particle to form a nitrogen nucleus. Represent the change by an equation showing the atomic and mass number of each elements.
Answer: When the nucleus of an element emits a beta particle, the number of protons increases by 1 and number of neutrons decreases by 1.

Question 62: Two radioactive nuclei are represented by xAp and yAq and when p and q are mass numbers and x, y are the atomic number. How can the products be represented, i .e., what are the new values of p, q. x and y after the emission of an α-particle and a β-particle from A and B respectively.
Answer:
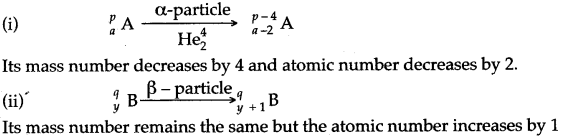
Question 63: A nucleus of an element has the symbol 84P202, and emits an α-particle and then a β-particle. The final nucleus is bQa Find a and b.
Answer:

Question 64: The nucleus B emit a β-particle and is transformed into nucleus C. What is the composition of C?
Answer: With the emission of a β-particle from nucleus B, number of protons will increase by one, i.e., number of protons = 82 + 1 = 83 and number of neutrons will be 126 – 1 = 125, i.e., the nucleus C will have 83 protons and 125 neutrons.
Mass number of nucleus A = Number of protons + number of neutrons
= 84 + 128 = 212.
Question 65: Explain briefly what changes take place within the nuclei when beta particles are emitted by a radioactive substance.
Answer: The mass number remains unchanged but atomic number increases by 1 of the nuclei undergoing such a change.
Question 66: Which part of the atom undergoes a change in the process of radioactive decay?
Answer: The nucleus of the atom undergoes a change in the process of radioactive decay.
Question 67: How do infrared and γ-rays differ in (i) wavelength, (ii) penetrating power?
Answer: (i) Infrared rays are of much longer wavelength than that of γ rays. The wavelength of infrared rays lies between 8000 Å to 107 Å while that of γ rays lies between 0.001 Å to 0.1 Å.
(ii) γ rays are most penetrating while infrared rays almost have negligible penetrating power.
Question 68: From α, β and γ-rays, name the one which travels with the speed of light?
Answer: γ-rays travel with the speed of light.
Question 69: From α, β and γ-rays, name the one which is not affected by electric or magnetic field?
Answer: γ-rays are not affected by electric or magnetic field.
Question 70: Arrange α, β and γ-rays in ascending order with respect to their (i) Penetrating power,
(ii) Ionizing power and (iii) Biological effect.
Answer: Given below:

Question 71: What changes occur in the nucleus of radioactive element when it emits gamma radiation? Give an example.
Answer: When the nucleus of a radioactive element emits gamma radiation, there is no change inside the nucleus except that its excess energy is released in the form of y-radiation.
![]()
The star indicates the excited state of nucleus.
Question 72: What is the value of the speed of gamma radiations in air or vacuum?
Answer: Speed of γ radiation = 3 × 108 m/s in air or vacuum.
Question 73: What are beta rays and gamma rays?
Answer: β-rays are a stream of electrons emitted by the nucleus of an atom and γ-rays are electromagnetic waves of high frequency given off by nuclei.
Question 74: Name the radioactive radiations which have the least penetrating power.
Answer: a-radiations have the least penetrating power.
Question 75: Explain why alpha and beta-particles are deflected in an electric or a magnetic field but gamma: rays are not deflected in such a field.
Answer: α and β are positive and negative charged particles respectively, therefore these are deflected in electric or magnetic field whereas γ radiations are not charged particles so does not deflect.
Question 76: A radioactive substance, is oxidised. What change would you expect to take place in the nature of radioactivity? Give reason for your answer.
Answer: No changes as the nucleus of the element is not involved.
Question 77: State the medical use of radioactivity.
Answer: (i) Radioactive radiations (gamma rays) are used to cure diseases such as cancer etc.
(ii) Radio isotopes such as NaCl etc. are used for diagnosis.
Question 78: explain, why radium paint, consisting of zinc sulphide and a trace of radium salt, glows in the dark?
Answer: Radium is a radioactive substance. It gives out radioactive radiations which show fluorescence in zinc sulphide. Zinc sulphide is a fluorescent material, so it glows in dark.
Question 79: State one use of radio-isotopes.
Answer: The most important one use of radio-isotopes is in medicines which are used mainly to cure cancer.
Question 80: State three safety precautions that you would take while using a radioactive substance.
Answer: (i) We should put on special lead lined aprons, lead gloves and special film badges.
(ii) We should not touch the radioactive substances but should handle them with long lead tongs.
(iii) The radioactive substance should be kept in a thick walled lead container:
Question 81: It is advised not to touch a radioactive substance by hand. Give reason.
Answer: The reason is that the radiations (α, β and γ) emitted by a radioactive substance have large penetrating power.
Question 82: Which of the radioactive radiations can cause severe genetical disorders and are deflected by an electric field?
Answer: γ-radiations can cause severe genetical disorder α and β are deflected by an electric field.
Question 83: Give any two important sources of background radiation.
Answer: Two sources of background radiations are:
(i) Potassium present in our body.
(ii) Cosmic rays coming from outer space.
Question 84: What is mean by background radiation?
Answer: Background radiations are the radioactive radiations to which we all are exposed even in the absence of an actual visible radioactive source. It total does is not very large, so it oes not cause any serious biological damage to us.
Question 85: What is nuclear energy? Name the process used for producing electricity using nuclear energy.
Answer: Nuclear energy is the energy released by the atom’s nucleus during a nuclear reaction. It is an exothermic process. The process of Nuclear fission is used to produce electricity.
Question 86: State one important advantage and disadvantage each of using nuclear energy for producing electricity.
Answer: Advantage:
Higher order of electrical energy can be produced.
Disadvantage: Difficult to control the higher order energy release as it damages the control rods and difficult to dispose the nuclear wastes.
Question 87: State the energy conversion taking place in a solar cell.
Answer: Energy conversion taking place in solar cell is Solar energy to Electric energy.
Question 88: What is meant by nuclear waste?
Answer: After disintegration the radioactive material finally converts into lead and still it holds some radioactivity. This is called nuclear waste.
Question 89: Suggest one effective way for the safe disposal of nuclear waste.
Answer: Delay and decay method is the effective way for the safe disposal of nuclear waste.
Long Answers
Question 1: State three ways of obtaining an electron-beam.
Answer: The three ways are:
(i) In a cathode ray tube, when pressure of 0.01 mm of Hg and a high p.d. of about 10000 V is applied across its electrodes.
(ii) In a diode-valve, it is produced from the heated cathode from where they are attracted by the anode plate kept at a high positive potential.
(iii) Electrons are also produced in the process known as ‘photo electric emission’ by focussing light on a metal surface coated with cesium oxide.
Question 2: Name the particles that constitute the cathode rays. State some properties of cathode rays.
Answer: Cathode rays are made up of electrons—the fundamental negatively charged particles. These rays have the following properties, among others.
(i) They can affect photographic plates and produce fluorescence in materials like zinc sulphide.
(ii) They carry momentum and energy.
(iii) They are deflected by electric and magnetic field in the sense in which negatively charged particles are deflected.
Question 3: Mention four properties of Becquerel rays.
Answer: The four properties of Becqueral rays are:
(i) They have penetrating power.
(ii) They affect a photographic plate.
(iii) They can ionize a gas or air through which they pass.
(iv) They are deflected by electric and magnetic fields.
Question 4: Name the three main parts of a hot cathode ray tube. Mention one important function of ear main parts:
State the principle on which the functioning of a nuclear reactor is based.
Answer: Three main parts of hot cathode ray tube:
(i) The electron gun,
(ii) The deflecting system,
(iii) The fluorescent screen.
Functions:
(i) Electron gun gives out fine beam of electrons.
(ii) The deflecting system deflect electron beam in horizontal and vertical direction.
(iii) The fluorescent screen gives bright spot on stirking of electron.
Question 5: What precautions must be taken for the safe disposal of nuclear waste?
Answer: The nuclear waste obtained from laboratories, hospitals, scientific establishments or power plants must be first kept in thick casks and then they should buried in the specially designed deep underground stores. The stores should be make quite far from the populated area and the casks should also be buried and scaled in useless mines after storing if.
Question 6: How many alpha and beta particles are emitted when Uranium nucleus 9U238 decays to Lead If 82Pb206 ?
Answer: On emission of one alpha particle, the mass number decreases by 4. With the emission of an alpha particle, the atomic number decreases by 2 but when a β-particle is emitted, the atomic number increases by 1.
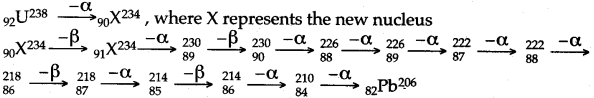
No. of α-particles emitted = 8.
No. of β-particles emitted = 6.
Question 7: State three safety precautions that you would take while handling the radioactive substances.
Answer: (i) The people working with radioactive materials should use mechanical devices such as tongs, clamps, tweezers, etc., to help minimize exposure.
(ii) The people working with radioactive materials should wear special lead lined lab coasts, gloves and safety glasses and remove them when leaving the work area or laboratory.
(iii) All radioactive materials should be stored in specially designed thick lead containers with a very narrow opening so as to stop radiations coming out from other directions.
Question 8: What safety measures and needed in a nuclear power plant?
Answer: (i) The nuclear power plant must be shielded with lead and steel walls so as to stop radiations from escaping out to the environment during its normal operations.
(ii) The nuclear power plant must be housed in an airtight building of strong concrete structure which can withstand earthquakes, fires and explosions.
(iii) There must be a back up of cooling system for the reactor core, so that in case of failure of one system, the other system could take it place.
Figure Based Short Answers
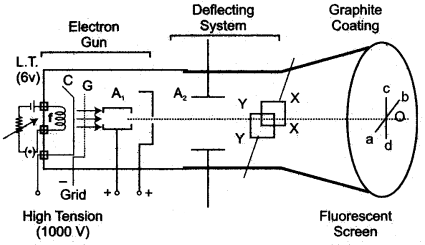
Question 1: Draw a simplified labelled diagram of a hot cathode ray tube.
Answer:
Question 2: The figure (i) shows a radioactive source S placed in a thick walled lead container. The radiations given off pass through a magnetic field acting in direction perpendicular to the plane of paper inwards as shown by X. Copy the diagram and show the path of radiations. Explain why the source is kept in a thick-walled lead container.
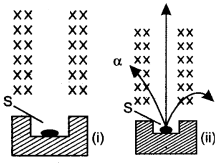
Answer: The figure (ii) shows the path of radiation in the magnetic field. The source is kept in a thick-walled lead container so that the it.
Radiations may not come out in the undesired direction otherwise they will harm the person working on.
Figure Based Long Answers
Question 1: Answer the questions with respect to the following diagram:
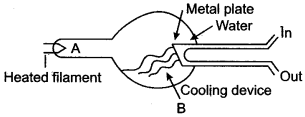
(i) Name the charged particles emitted from the heated filament? Name the phenomenon of emission of these particles.
(ii) A beam of such particles, after acceleration is allowed to strike a metal plate C. Radiation B is given off. Name the radiation B.
(iii) Name the material of target C.
(iv) State two properties of radiation B different from those of visible light.
(v) State two factors on which the wavelength of radiation B depends.
(vi) State two uses of radiation B.
(vii)Why is it necessary to cool the tube?
Answer: (i) ‘Electrons’. The phenomenon of emission of these particles is called “thermionic emission”.
(ii) Radiation B is called “X-rays”.
(iii) The material of the target C is “tungsten.”
(iv) The properties of radiation B differ from those of visible light are:
(a) These radiations can penetrate through the substances which are opaque to visible light.
(b) They destroy the living cells.
(v) The wavelength of radiation B depends upon:
(a) Potential difference applied between cathode and anode.
(b) Material of the target.
(vi) Uses:
(a) In the field of medical science (X-rays photography used in surgery)
(b) In scientific work (to study crystal structure).
(vii) A large amount of heat is produced when the high kinetic energy electrons strike the target. Therefore it is necessary to cool the tube.
Question 2: A certain nucleus X has a mass number 14 and atomic number 6. The nucleus X changes to 7Y14 after the loss of a particle.
(i) Name the particle emitted.
(ii) Represent this change in the form of an equation.
(iii) A radioactive substance is oxidized. What change would you expect to take place in the nature of its radioactivity? Give a reason for your answer.
Answer: (i) Particle emitted is β-particle.
(ii) 6X14 → 7Y14 + -1β0
(iii) No change. Since the phenomenon of radioactivity is not due to the orbital electrons which would be easily affected by such changes. Radioactivity is the property of the nucleus.
Short Numericals
Question 1: An element P disintegrates by beta-emission and the new element suffers two further disintegration, both by beta-emission, to form an element Q. Show that P and Q are isotopes.
Solution:

The atomic numbers (Z) of P and Q are the same but their mass numbers [A and (A-4)] are different. So P and Q are isotopes.
Question 2: An element X has the symbol 84X202. It emits an alpha-particle and then a beta-particle. The final nucleus is bYa. Find a and b.
Solution:
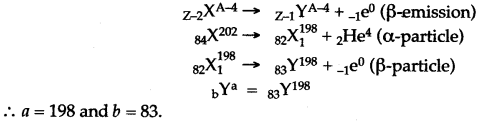
Question 3: Complete the following fission reaction
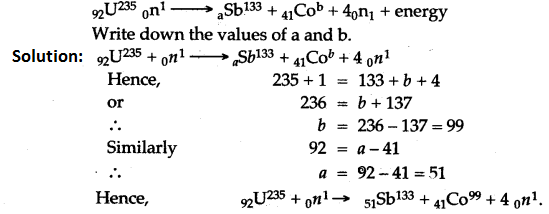
Long Numericals
Question 1: A certain nucleus X has a mass number 15 and atomic number 7. Find the number of neutrons. The nucleus X losses (i) one proton (ii) one β-particle, (iii) one α-particle, Express each change and write the symbol of the new nucleus in each case.
Solution:

Question 2: An element 92U238 losses one alpha particle, and then one β-particle and then again one β- particle to get the final nucleus bQa.
(i) Write down the values of a and b.
(ii) State the relationship between the nuclei P and Q.
Solution:

Question 3: An isotope of uranium is 92Y238, i.e., it has mass number 235 and atomic number 92.
(i) Find the number of electrons in the neutral atom of this isotope.
(ii) Find the number of protons and neutrons in its nucleus.
(iii) Do all isotopes have the same number of neutrons?
(iv) What is the number of protons in 92U238?
(v) Mention the relationship between 92U235 and 92U238.
Solution:

For More Resources