How are the Anode and Cathode rays Produced?
Atoms are made up of three subatomic particles electrons, Protons and neutrons. Electron has negative charge, proton has positive charge, whereas neutron has no charge, it is neutral. Protons and neutrons are present in a small nucleus at the centre of the atom. Electrons are outside the nucleus. The atoms of different elements differ in the number of electrons, protons and neutrons.
Thermionic Emission:
- Thermionic emission is a process of emission of electrons from the surface of a heated metal.
- Figure (a) shows the surface of a metal containing a large number of electrons which are free to move. The electrons are free to move at the surface but remain inside the metal.
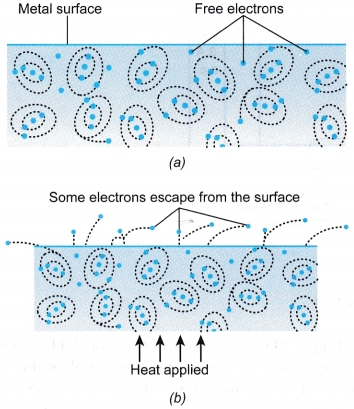
- The electrons at the surface cannot escape because they are held back by the attractive forces of the atomic nuclei near the surface.
- If the metal is heated to a high temperature as shown in Figure (b), some of the free electrons may gain sufficient energy to escape from the metal.
- Electrons are more easily emitted if the metal is coated with an oxide of barium or strontium.
- The rate of thermionic emission depends on:
(a) Surface area of the metal
The larger the surface area, the more the number of electrons emitted.
(b) Temperature of the metal
The higher the temperature, the more the number of electrons which will gain enough energy to escape from the metal surface.
(c) Type of metal
Different types of metal requires different amounts of energy for the electrons to escape.
(d) Nature of the metal surface
When the metal surface is coated with metal oxide, electrons can be emitted at a lower temperature.
How are the cathode rays produced?
- Thermionic emission is used to produce a continuous supply of electrons. These electrons can be made to move at high speed in a vacuum tube as shown in Figure.
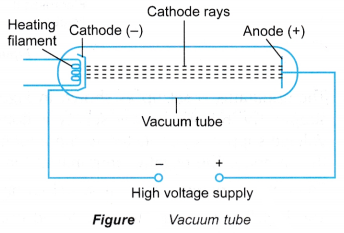
- The vacuum tube is an evacuated glass tube where streams of electrons can move without any obstruction by air molecules.
- Inside the vacuum tube there are positive and negative electrodes. The positive electrode is called anode and the negative electrode is called cathode. When a high voltage is applied between the cathode and the anode, the electrons are accelerated at high speed from the cathode to the anode. This stream of electrons which is moving at high speed is called cathode rays.
People also ask
- What is meant by cathode ray oscilloscope?
- Cathode Ray Oscilloscope Construction and Working
- What are the functions of an oscilloscope?
- What is the use of cathode ray oscilloscope?
- Cathode Ray Oscilloscope Experiment
Properties of Cathode Rays
- Cathode rays always travel in straight line.
- Cathode rays consist of material particles and possess energy, hence they can produce mechanical effects.
- Cathode rays consist of negatively charged particles.
- Cathode rays can penetrate through thin metalic sheets.
- Cathode rays ionize the gas through which they travel.
- Cathode rays heat up the object on which they fall. When they strike an object, a part of the kinetic energy is transferred to the object resulting in a rise in temperature.
- Cathode rays produce a green fluorescence on glass surface.
- When cathode rays fall on certain metals like copper, X-rays are produced. X-rays are not deflected by any electrical or magnetic fields but they pass through the opaque material and are only stopped by solid objects like bones.
- The mass of electrons is very small compared to the mass of an atom. The same type of negatively charged particles are formed even if different gases are taken in the discharge tube or different metals are used as cathode.
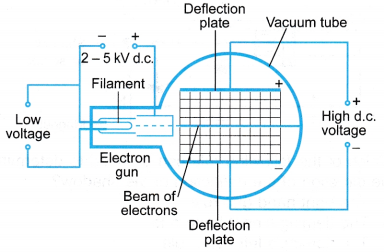
The properties of the cathode rays can be studied using an apparatus such as the Maltese Cross tube as shown in Figure.

- When the filament in the cathode is connected to a low voltage power supply, the light from the filament will cast a shadow of the Maltese Cross on the screen directly behind the cross. This is due to the property of light travelling in a straight line.
- When the E.H.T. power supply connected across the cathode and the anode is switched on, the shadow cast by the Maltese Cross becomes darker. The screen around the shadow emits green light.
- The green light around the shadow is caused by the cathode rays, which are not blocked by the Maltese Cross, accelerating down the tube and striking the fluorescent screen. This shows that cathode rays also travel in a straight line.
- The kinetic energy of the electrons is converted into light energy by a process called fluorescence. This takes place in the phosphor coating inside the end of the tube. This shows that cathode rays have energy and momentum.
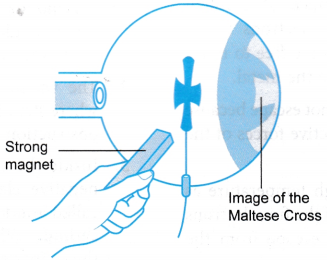
- When a bar magnet is brought to the neck of the tube, where the electrons emerge from the anode, two shadows are seen on the screen. The shadow cast by light is not affected by the magnet and stays at the centre. The shadow cast by the cathode rays is moved and distorted by the magnet. This shows that cathode rays are deflected by magnetic field.
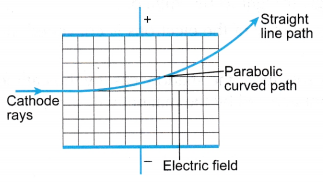
- When a cathode ray deflection tube is connected as shown in Figure, the cathode rays are seen deflected towards the positive plate and away from the negative plate.
- The path of the cathode rays in an electric field is the shape of a parabola as shown in Figure. This shows that cathode rays are negatively charged.
- The properties of the cathode rays can be summarised as follow:
(a) These rays originate from the cathode; therefore they are beams of negatively charged particles called electrons.
(b) They travel in straight lines and cast sharp shadows.
(c) They travel at high speed and have kinetic energy and momentum.
(d) They can do work and therefore can generate fluorescence when they hit the fluorescent screen.
(e) They are deflected by magnetic fields and electric fields.
Properties of Cathode Rays Experiment
Aim: To study the properties of cathode rays.
Materials: Strong magnet, connecting wires
Apparatus: Maltese Cross tube, Extra High Tension (E.H.T.) power supply, 6 V a.c. power supply
Method: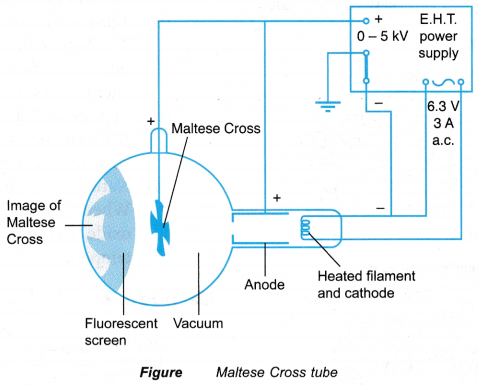
- The apparatus is set up as shown in Figure by connecting a Maltese Cross tube to the E.H.T. power supply. The 6 V a.c. power supply is connected to the heating filament. The positive terminal of the high voltage supply is connected to the anode and Maltese Cross, while the negative terminal is connected to the cathode. The negative terminal of the E.H.T. supply is earthed.
- The 6 V a.c. power supply is switched on and the shadow formed on the screen is observed.
- The E.H.T. power supply is switched on. The voltage is increased slowly from zero to above 2 kV. The shadow and the colour on the screen is observed.
- A strong magnet is placed at one side of the Maltese Cross tube as shown in Figure. What happens to the shadow is observed.
Observations:
- When the 6 V a.c. power supply is switched on, a dark shadow of the Maltese Cross is formed on the screen.
- When the E.H.T. power supply is switched on, a darker shadow of the Maltese Cross is seen on the screen. The shadow of the Maltese Cross is surrounded by a green light.
- When a strong magnet is placed at one side of the Maltese Cross tube, two shadows are seen on the screen. The lighter shadow remains at the centre of the screen, the darker one is shifted downwards and is distorted.
Discussion:
- The dark shadow of the Maltese Cross is due to the light from the filament when it is switched on.
- The darker shadow shown on the screen when the E.H.T. power supply is switched on is due to the stream of cathode rays blocked by the Maltese Cross. The stream of electrons that are not blocked when travelling down the tube strikes the fluorescent screen and causes a green light to be emitted.
- When the electrons strike the fluorescent screen, the kinetic energy of the electrons is converted into light energy.
- When a strong magnet is placed at the side of the Maltese Cross tube, the shadow cast by the cathode rays is moved and distorted. The shadow cast by light which is not affected by the magnet will stay at the centre of the screen.
How are the anode rays produced?
The pressure in the tube is decreased, it was observed that in addition to cathode rays, a new kind of rays are also found which came through the perforations (holes) of the cathode. These rays travelled in opposite direction of the cathode rays and passed through the holes of the cathode and struck the other end of the discharge tube. When these radiations struck the end of the tube P, the fluorescent radiations were also produced. These rays were called canal rays because they are passed through the holes or canals in the cathode. These were also called anode rays because they move from the anode. It was found that the anode rays consist of positively charged particles. Therefore, these rays were also called positive rays.
Properties of Anode Rays
- Anode rays travel in straight lines.
- Anode rays consist of material particles.
- Anode rays are deflected by electric field towards negatively charged plate. This indicates that they are positively charged.
- Anode rays are deflected by magnetic field. The direction of deflection indicates that they are positively charged.
- The charge to mass ratio of the particles in the anode rays was determined by W. Wien by using Thomson’s technique. Charge to mass ratio of the particles in the anode rays depends upon nature of the gas taken in the discharged tube.
- The nature of anode rays depend on the nature of the gas taken in discharge tube.
