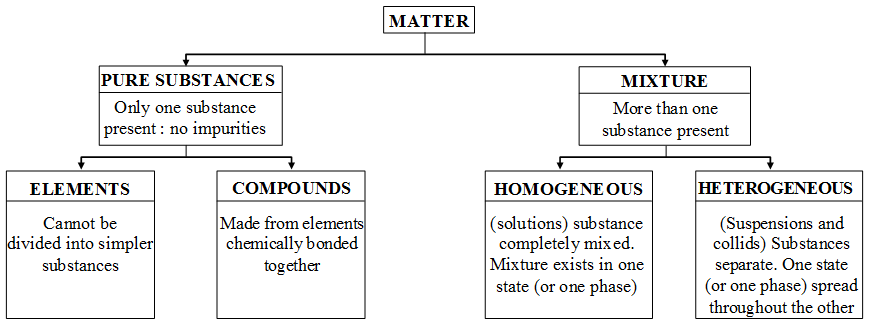
What are the types of Pure substances and Mixtures
All the matter around us is not pure. The matter around us is of two types.
In the previous chapter, we have learnt about the three states of matter. Before, understanding the chemical nature of matter, let us first understand the scientific meaning of the term chemical substance.
The scientific meaning of the term chemical substance is different from its everyday meaning. In terms of science, substance is a kind of matter that cannot be separated into other kinds of matter by any physical process. In other words, substance is a pure form of matter and not a mixture of several different kinds of matter. Most of the things that we use in our day-to-day life are in the form of mixtures. Pure substances are rare. For example, the dissolved sugar can be separated from its solution by some physical process (evaporation or distillation). However, sugar is itself a substance and cannot be separated by physical processes into its constituents. Similarly, common salt (sodium chloride), iron, mercury, calcium oxide, hydrochloric acid are substance.
Pure substances
- A pure substance consists of a single type of particles.
- Pure substances are always homogeneous.
- All the elements and compounds are pure substances because they contain only one kind of particles.
- A pure substance cannot be separated into other kinds of matter by any physical process.
- A pure substance has a fixed composition as well as a fixed boiling point and melting point.
Ex. Hydrogen, Oxygen, Copper, Gold, Silver.
Pure substances can be divided into two types.
Mixtures
A mixture is a substance which consists of two or more elements or compounds not chemically combined together. All the solutions are mixtures. The various substances present in a mixture are known as “constituents of the mixture” or “components of the mixture”.
Ex. Lemonade (nimbu pani) is a mixture of water, lemon juice, sugar and salt.
A mixture consists of two or more different type of particles having different chemical nature. Mixture may be homogeneous or heterogeneous. All the mixtures are impure substances. A mixture does not have a fixed composition or a fixed melting point and boiling point.
Types of mixtures
Mixtures are of two types :
