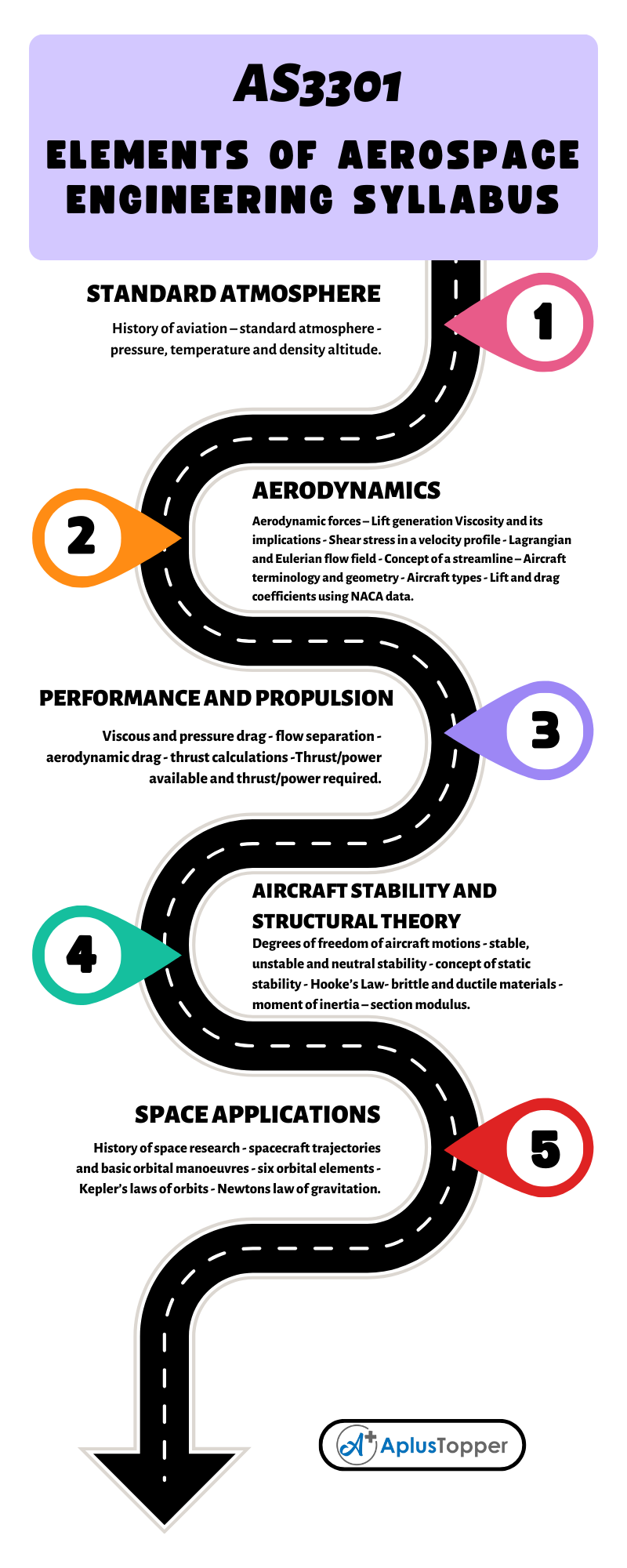
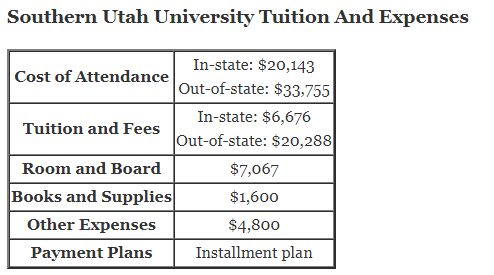
Oxidation and Reduction in Chemical Cells
Oxidation and Reduction in Chemical Cells In a simple voltaic cell, two different metals are in contact with an electrolyte. The more electropositive metal will become the negative terminal while the less electropositive metal will become the positive te…