ICSE Chemistry Previous Year Question Paper 2016 Solved for Class 10
ICSE Paper 2016
CHEMISTRY
(Two Hours)
Answers to this Paper must be written on the paper provided separately.
You will not be allowed to write during the first 15 minutes.
This time is to be spent in reading the Question Paper.
The time given at the head of this Paper is the time allowed for writing the answers.
Section I is compulsory. Attempt any four questions from Section II.
The intended marks for questions or parts of questions are given in brackets [ ].
SECTION-I (40 Marks)
(Attempt all questions from this Section)
Question 1:
(a) Fill in the blanks with the choices given in brackets. [5]
- Metals are good ………. (oxidizing agents/reducing agents) because they are electron ……….. (acceptors/donors).
- Electrovalent compounds have ………… (high/low) melting points.
- Higher the pH value of a solution, the more ……….. (acidic/alkaline) it is.
- ………. (AgCl/PbCl2) white precipitate is soluble in excess NH4OH.
- Conversion of ethene to ethane is an example of ……….. (hydration/hydrogenation).
(b) Choose the correct answer from the options given below: [5]
- An element with the atomic number 19 will most likely combine chemically with the element whose atomic number is:
(A) 17 (B) 11 (C) 18 (D) 20 - The ratio between the number of molecules in 2g of hydrogen and 32g of oxygen is:
(A) 1 : 2 (B) 1 : 0.01 (C) 1 : 1 (D) 0.01 : 1
[Given that H = 1, O = 16] - The two main metals in Bronze are:
(A) Copper and zinc (B) Copper and lead
(C) Copper and nickel (D) Copper and tin - The particles present in strong electrolytes are:
(A) Only molecules (B) Mainly ions
(C) Ions and molecules (D) Only atoms - The aim of the Fountain Experiment is to prove that:
(A) HCl turns blue litmus red
(B) HCl is denser than air
(C) HCl is highly soluble in water
(D) HCl fumes in moist air.
(c) Write balanced chemical equations for each of the following: [5]
- Action of warm water on AIN.
- Action of hot and concentrated Nitric acid on copper.
- Action of Hydrochloric acid on sodium bicarbonate.
- Action of dilute Sulphuric acid on Sodium Sulphite.
- Preparation of ethanol from Ethyl Chloride.
(d) State your observations when: [5]
- Dilute Hydrochloric acid is added to Lead nitrate solution and the mixture is heated.
- Barium chloride solution is mixed with Sodium Sulphate Solution.
- Concentrated Sulphuric acid is added to Sugar Crystals.
- Dilute Hydrochloric acid is added to Copper carbonate.
- Dilute Hydrochloric acid is added to Sodium thiosulphate.
(e) Identify the term/substance in each of the following: [5]
- The tendency of an atom to attract electrons to itself when combined in a compound.
- The method used to separate ore from gangue by preferential wetting.
- The catalyst used in the conversion ofethyne to ethane.
- The type of reactions alkenes undergo.
- The electrons present in the outermost shell of an atom.
(f) (i) A gas of mass 32gms has volume of 20 litres at S.T.P. Calculate the gram molecular weight of the gas.
(ii) How much Calcium oxide is formed when 82g of calcium nitrate is heated? Also find the volume of nitrogendioxide evolved:
2Ca(NO3)2 → 2CaO + 4NO2 + O2
(Ca = 40, N = 14, O = 16) [5]
(g) Match the salts given in Column I with their method of preparation given in Column II: [5]

(h) (i) Write the IUPAC names of each of the following: [5]
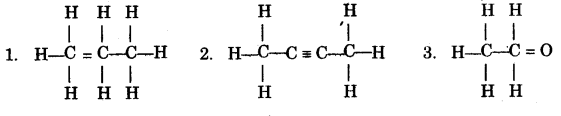
(ii) Rewrite the following sentences by using the correct symbol > (greater than) or < (less than) in the blanks given:
- The ionization potential of Potassium is ………. that of Sodium.
- The electronegativity of Iodine is ………… that of Chlorine.
Answer:
(a)
- Reducing agents, donors
- High
- Alkaline
- AgCl
- Hydrogenation
(b)
- (A) 17
- (C) 1 : 1
- (D) Copper and tin
- (B) Mainly ions
- (C) HCl is highly soluble in water.
(c)
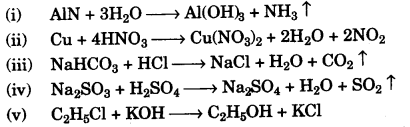
(d)
- A white precipitate of lead chloride appears which dissolves on heating.
- A white insoluble precipitate of Barium sulphate is obtained.
- White crystals of sugar turn to a black spongy mass called sugar charcoal.
- A brisk effervescence with the release of colourless, odourless, acidic gas that extinguish glowing splint i.e., carbon dioxide gas is evolved.
- A colourless gas with smell of burning sulphur i.e., Sulphur dioxide is released. A yellow crystal settles at the bottom i.e., sulphur.
(e)
- Electron affinity
- Froth Floatation Method (concentration of ore)
- Nickel
- Addition reaction
- Valence electron
(f)


(g)

(h) (i)
- Prop-1-ene
- Butr-2-yne
- Ethan-1-al
(ii)
- < (less than)
- < (less than)
SECTION-II (40 Marks)
(Attempt any four questions from this section)
Question 2:
(a) Use the letters only written in the Periodic Table given below to answer the questions that follow: [4]
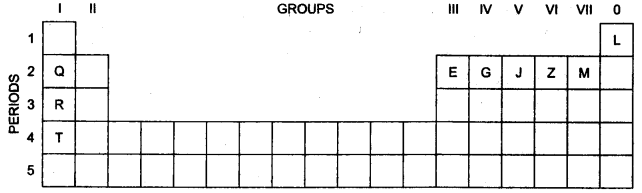
- State the number of valence electrons in atom J.
- Which element shown forms ions with a single negative charge?
- Which metallic element is more reactive than R?
- Which element has its electrons arranged in four shells?
(b) Fill in the blanks by selecting the correct word from the brackets: [2]
- If an element has a low ionization energy then it is likely to be …………. (metallic/non metallic).
- If an element has seven electrons in its outermost shell then it is likely to have the ……….. (largest/smallest) atomic size among all the elements in the same period.
(c) The following table shows the electronic configuration of the elements W, X, Y, Z:
| Element | W | X | Y | Z |
| Electronic configuration | 2, 8, 1 | 2, 8, 7 | 2, 5 | 1 |
Answer the following questions based on the table above:
- What type of Bond is formed between: [2]
1. W and X 2. Y and Z - What is the formula of the compound formed between: [2]
1. X and Z 2. W and X
Answer:
(a)
- No. of Valence electrons in atom J = 5
- Element M
- Element T is more reactive than R.
- Element T has its electrons arranged in four shells
(b)
- Metallic
- Smallest
(c) (i)
- Ionic bond.
- Covalent bond.
(ii)
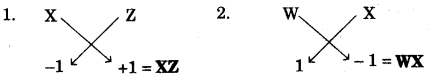
Question 3:
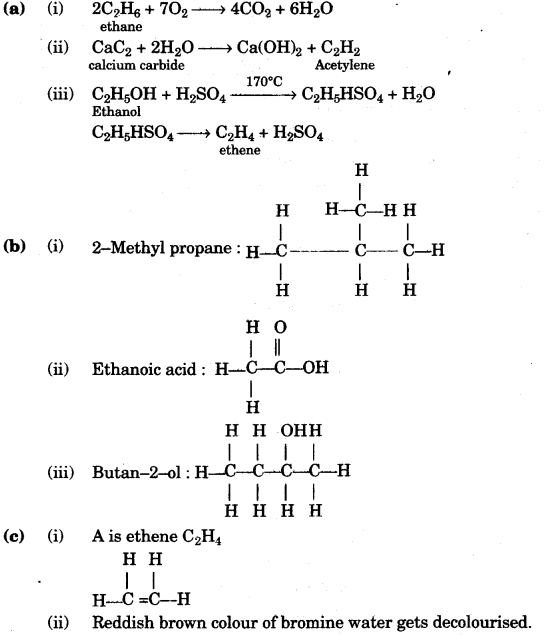
(a) Write a balanced chemical equation for each the following: [3]
- Burning of ethane in plentiful supply of air.
- Action of water on Calcium carbide.
- Heating of Ethanol at 170°C in the presence of conc. Sulphuric acid.
(b) Give the structural formulae of each of the following: [3]
- 2-methyl propane
- Ethanoic acid
- Butan-2-ol
(c) Equation for the reaction when compound A is bubbled through bromine dissolved in carbon tetrachloride is as follows: [2]
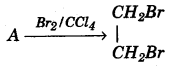
- Draw the structure of A.
- State your observation during this reaction.
(d) Fill in the blanks using the appropriate words given below: [2]
(Sulphur dioxide, Nitrogen dioxide, Nitric oxide, Sulphuric acid)
- Cold, dilute nitric acid reacts with copper to give ………..
- Hot, concentrated nitric acid reacts with sulphur to form ……….
Answer:
(d)
- Nitric oxide.
- Sulphuric acid
Question 4:
(a) Identify the gas evolved and give the chemical test in each of the following case: [2]
- Dilute hydrochloric acid reacts with sodium sulphite.
- Dilute hydrochloric acid reacts with iron (II) sulphide.
(b) State your observations when ammonium hydroxide solution is added drop by drop and then in excess to each of the following solutions: [2]
- copper sulphate solution.
- zinc sulphate solution.
(c) Write equations for the reactions taking place at the two electrodes (mentioning clearly the name of the electrode) during the electrolysis of: [4]
- Acidified copper sulphate solution with copper electrodes.
- Molten lead bromide with inert electrodes.
(d)
- Name the product formed at the anode during the electrolysis of acidified water using platinum electrodes.
- Name the metallic ions that should he present in the electrolyte when an article made of copper is to be electroplated with silver. [2]
Answer:
(a)
- Sulphur-di-oxide gas is evolved.
Colourless, gas with smell of burning sulphur. It turns acidified potassium dichromate orange to green. - Hydrogen sulphide gas is evolved.
Colourless gas with smell of rotten eggs. It turns lead acetate paper black.
(b)
- Bluish white precipitate appears which dissolves in excess of NH4OH and give an inky blue solution of tetra amino copper sulphate.
- A gelatinous white precipitate appears which dissolves in excess of NH4OH and give a clear transparent colourless solution of tetra amine zinc sulphate.
(c)
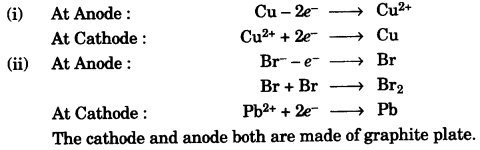
(d)
- Oxygen gas.
- Silver ions/Ag+.
Question 5:
(a) A gas cylinder contains 12 × 1024 molecules of oxygen gas.
If Avogadro’s number is 6 × 1023; Calculate:
(i) the mass of oxygen present in the cylinder.
(ii) the volume of oxygen at S.T.P. present in the cylinder. [O = 16] [2]
(b) A gaseous hydrocarbon contains 82.76% of carbon. Given that its vapour density is 29, find its molecular formula. [C = 12, H = 1] [3]
(c) The equation 4NH3 + 5O2 → 4NO + 6H2O, represents the catalytic oxidation of ammonia. If 100 cm3 of ammonia is used calculate the volume of oxygen required to oxidise the ammonia completely. [3]
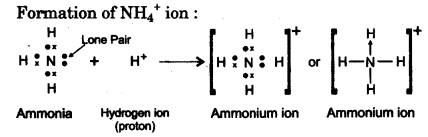
(d) By drawing an electron dot diagram show the formation of Ammonium Ion [AtomicNo. :N =7 and H = 1] [2]
Answer:
(a)
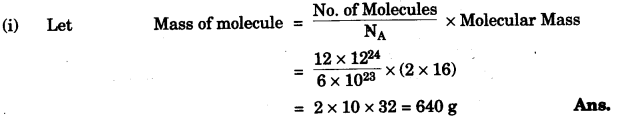
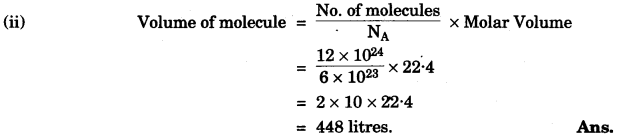
(b)

(c)
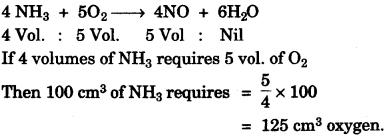
(d)
Question 6:
(a) Name the gas evolved when the following mixtures are heated: [2]
- Calcium hydroxide and Ammonium Chloride.
- Sodium Nitrite and Ammonium Chloride.
(b) Write balanced chemical equations for each of the following: [2]
(i) When excess of ammonia is treated with chlorine.
(ii) An equation to illustrate the reducing nature of ammonia.
(c) A, B, C and D summarize the properties of sulphuric acid depending on whether it is dilute or concentrated. [3]
A = Typical acid property
B = Non volatile acid
C = Oxidizing agent
D = Dehydrating agent
Choose the property (A, B, C or D) depending on which is relevant to each of the following:
- Preparation of Hydrogen chloride gas.
- Preparation of Copper sulphate from copper oxide.
- Action of cone. Sulphuric acid on Sulphur.
(d) Give reasons why: [3]
- Sodium Chloride will conduct electricity only in fused or aqueous solution state.
- In the electroplating of an article with silver, the electrolyte sodium argento-cyanide solution is preferred over silver nitrate solution.
- Although copper is a good conductor of electricity, it is a non-electrolyte.
Answer:
(a)
- Ammonia gas.
- Nitrogen gas.
(b)
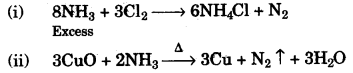
This reaction show the reducing nature of ammonia. NH3 reduces CuO into Cu.
(c)
- B—Non volatile acid.
- A—Typical acid property.
- C—Oxidizing agent.
(d)
- In solid state, sodium chloride does not contain free ions so it will not conduct electricity but in fused or aqueous state it contains free mobile ions that conducts electricity.
- Silver nitrate solution undergoes rapid dissociation that can cause non-uniform coating therefore it is not preferred. Whereas sodium argentocyanide to cyanide solution is a complex salt and undergoes slow decomposition and ensure smooth and uniform coating.
- Copper is a metals so it conducts electricity by flow of electrons but it does not form ions so it is a non electrolyte.
Question 7:
(a) (i) Name the solution used to react with Bauxite as a first step in obtaining pure aluminium oxide, in the Baeyer’s process.
(ii) Write the equation for the reaction where the aluminium oxide for the electrolytic extraction of aluminium is obtained by heating aluminium hydroxide.
(iii) Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
(iv) Write the equation for the reaction that occurs at the cathode during the extraction of aluminium by electrolysis.
(v) Explain why it is preferable to use a number of graphite electrodes as anode instead of a single electrode, during the above electrolysis. [5]
(b) State what would you observe when:
- Washing Soda Crystals are exposed to the atmosphere.
- The salt ferric chloride is exposed to the atmosphere. [2]
(c) Identify the cations in each of the following case:
- NaOH solution when added to the Solution (A) gives a reddish brown precipitate.
- NH4OH Solution when added to the Solution (B) gives white ppt which does not dissolve in excess.
- NaOH Solution when added to Solution (C) gives white ppt which is insoluble in excess. [3]
Answer:
(a)
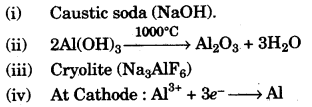
(v) The gas released at the anode is oxygen which will react with the heated graphite rod and form carbon dioxide and gradually get exhausted. Taking a number of graphite rods will compensate the loss and allow the process to go on for a longer time.
(b)
- Washing soda crystals are efflorescent substance. When they are exposed to atmosphere, they lose their water of crystallisation and become an amorphous powder.
- Ferric chloride is a deliquescent substance. When they are exposed to atmosphere, it will absorb water vapour and dissolve in it.
(c)
- Ferric ion/Fe3+.
- Lead ion/Pb2+.
- Calcium ions/Ca2+.
