ICSE Chemistry Previous Year Question Paper 2010 Solved for Class 10
ICSE Paper 2010
CHEMISTRY
(Two Hours)
Answers to this Paper must be written on the paper provided separately.
You will not be allowed to write during the first 15 minutes.
This time is to be spent in reading the Question Paper.
The time given at the head of this Paper is the time allowed for writing the answers.
Section I is compulsory. Attempt any four questions from Section II.
The intended marks for questions or parts of questions are given in brackets [ ].
SECTION-I (40 Marks)
(Attempt all questions from this Section)
Question 1:
(a) From the list given below, select the word(s) required to correctly complete the blanks (1) to (5) in the following passage:
Note: words chosen from the list are to be used only once. Write only the answers. Do not copy the passage.
[reddish brown, ammonium, nitrogen dioxide, hydroxyl, dirty green, ammonia, acidic, alkaline]
Nitrogen and hydrogen combine in the presence of a catalyst to give (1) ………… gas. When the above mentioned gas is passed through water it forms a solution which will be (2) ………….. in nature and the solution contains (3) …………. ions and (4) ………….. ions. The above solution when added to iron(II) sulphate solution, give a (5) …………… coloured precipitate of iron(II) hydroxide. [5]
(b) Select from the list given (A to E) one substance in each case which matches the description given in parts (1) to (5). (Note: Each substance is used only once in the answer.)
(A) Nitroso Iron(II) sulphate (B) Iron(III) chloride (C) Chromium sulphate (D) Lead(II) chloride (E) Sodium chloride.
- A compound which is deliquescent.
- A compound which is insoluble in cold water, but soluble in hot water.
- The compound responsible for the brown ring during the brown ring test of nitrate ion.
- A compound whose aqueous solution is neutral in nature.
- The compound which is responsible for the green coloration when sulphur dioxide is passed through acidified potassium dichromate solution. ** [5]
** Answer has not given due to out of present syllabus.
(c) For part (c). 1 – (c). 10, select the correct answer from the choices A, B, C and D which are given.
Write only the letter corresponding to the correct answer. [10]
- A particular solution contains molecules and ions of the solute so it is a:
(A) weak acid (B) strong acid
(C) strong base (D) salt solution - A compound which liberates reddish brown gas around the anode during electrolysis in its molten state is:
(A) Sodium chloride (B) Copper(II) oxide
(C) Copper(II) sulphate (D) Lead(II) bromide - An organic compound undergoes addition reactions and gives a red colour precipitate with ammoniacal cuprous chloride. Therefore, the organic compound could be:
(A) Ethane (B) Ethene
(C) Ethyne (D) Ethanol - An organic weak acid is:
(A) Formic acid (B) Sulphuric acid
(C) Nitric acid (D) Hydrochloric acid - During ionization metals lose electrons, this change can be called:
(A) Oxidation (B) Reduction
(C) Redox (D) Displacement - Which one of the following is not true of metals:
(A) Metals are good conductors of electricity
(B) Metals are malleable and ductile
(C) Metals from non-polar covalent compounds
(D) Metal will have 1 or 2 or 3 electrons in their valence shell. - An example of a complex salt is:
(A) Zinc sulphate
(B) Sodium hydrogensulphate
(C) Iron(II) ammonium sulphate
(D) Tetrammine copper(II) sulphate - Aqua regia is a mixture of:
(A) Dilute hydrochloric acid and concentrated nitric acid
(B) Concentrated hydrochloric acid and dilute nitric acid
(C) Concentrated hydrochloric acid [1 partj and concentrated nitric acid [3 parts]
(D) Concentrated hydrochloric acid [3 parts] and concentrated nitric acid [1 part] - The organic compound mixed with ethanol to make it spurious is:
(A) Methanol (B) Methanoic acid
(C) Methanal (D) Ethanoic acid - The number of electrons present in the valence shell of a halogen is:
(A) 1 (B) 3
(C) 5 (D) 7
(d) State your observation for the following cases: [5]
- Moist blue litmus is introduced into a gas jar of sulphur dioxide. **
- Dry red rose petals are placed in the jar of sulphur dioxide. **
- Paper soaked in potassium permanganate solution is introduced into a gas jar of sulphur dioxide. **
- Ammonia gas is burnt in an atmosphere of oxygen in the absence of a catalyst.
- Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle.
** Answer has not given due to out of present syllabus.
(e) Match the column A with column B. [5]
| Column A | Column B |
| 1. Sodium chloride | Increases |
| 2. Ammonium ion | Covalent bond |
| 3. Electronegativity across the period | Ionic bond |
| 4. Non metallic character down the group | Covalent and Coordinate bond |
| 5. Carbon tetrachloride | Decreases |
Answer as follows:
(i) correct item from B matching sodium chloride.
(ii) correct item from B matching ammonium ion, and so on.
(f) Write the equation for each of the following reactions: [5]
(i) Sulphur is heated with concentrated sulphuric acid.
(ii) Zinc oxide is treated with sodium hydroxide solution.
(iii) Ammonium chloride is heated with sodium hydroxide.
(iv) Concentrated sulphuric acid is poured over sugar.
(v) Magnesium sulphate solution is mixed with barium chloride solution.
(g) (i) LPG stands for liquefied petroleum gas. Varieties of LPG are marketed including a mixture of propane (60%) and butane (40%). If 10 litre of this mixture is burnt, find the total volume of carbon dioxide gas added to the atmosphere. Combustion reactions can be represented as: [5]
![]()
(ii) Calculate the percentage of nitrogen and oxygen in ammonium nitrate. [Relative molecular mass of ammonium nitrate is 80, [H = 1, N = 14, O = 16]
Answer:
(a)
- Ammonia
- Alkaline
- Ammonium
- Hydroxyl
- Dirty green.
(b)
- (B) Iron(III) chloride
- (D) Lead(II) chloride
- (A) Nitroso Iron(II) sulphate
- (E) Sodium chloride
(c)
- (A)
- (D)
- (C)
- (A)
- (A)
- (C)
- (D)
- (D)
- (A)
- (D)
(d)
4. It burns with a yellowish green flame producing water vapour and nitrogen.
5. It produce dense white fumes of ammonium chloride.
(e)
- Sodium chloride – Ionic bond.
- Ammonium ion – Covalent and Co-ordinate bond.
- Electronegativity across a period – Increases.
- Non metallic character down the group – Decreases.
- Carbon tetrachloride – Covalent bond.
(f)
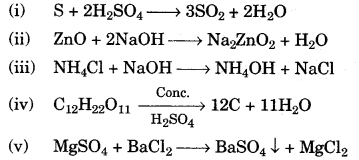
(g)


SECTION-II (40 Marks)
(Answer any four questions from this section)
Question 2:
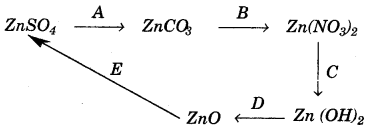
(a) Given the equations for the following conversions A to E.
(b) The questions below are related to the manufacture of ammonia:
(i) Name the process.
(ii) In what ratio must the reactants be taken?
(iii) Name the catalyst used.
(iv) Give the equation for the manufacture of ammonia.
(v) Ammonia can act as a reducing agent – write a relevant equation for such a reaction. [5]
Answer:
(a)
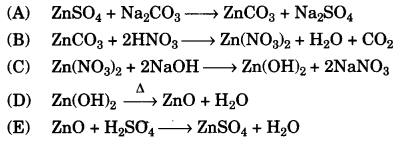
(b)
(i) Haber’s process.
(ii) 1 part of nitrogen gas and 3 parts of hydrogen gas.
(iii) Finely divided iron (Fe).
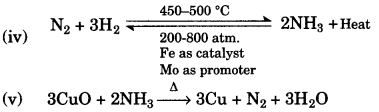
Question 3:
(a) Draw the structural formula for each of the following:
(i) Ethanoic acid (ii) But-2-yne. [2]
(b) Mr. Ramu wants to electroplate his key chain with nickel to prevent rusting. For this electroplating:
(i) Name the electrolyte
(ii) Name the cathode
(iii) Name the anode
(iv) Give the reaction at the cathode
(v) Give the reaction at the anode. [5]
(c) Three different electrolytic cells A, B and C are connected in separate circuits. Electrolytic cell A contains sodium chloride solution. When the circuit is complete a bulb in the circuit glows brightly. Electrolytic cell B contains acetic acid solution and in this case the bulb in the circuit glows dimly. The electrolytic cell C contains sugar solution and the bulb does not glow. Give a reason for each of these observations. [5]
Answer:
(a)

(b)
(i) Aqueous solution of Nickel Sulphate with few drops of dil. Sulphuric acid.
(ii) The key chain.
(iii) Pure Nickel bar.
(iv) Ni++ + 2e– → Ni
(v) Ni – 2e– → Ni++
(c)
A is sodium chloride solution which is a strong electrolyte which contains only ions. So more electricity is conducted and bulb glows brightly.
B is acetic acid which contains both ions and molecules that is less number of ions. So less electricity is conducted and bulb glows dimly.
C is sugar solution which is a non electrolyte that is it does not contain ions so electricity is not conducted and bulb does not glow.
Question 4:
(a) 4.5 moles of calcium carbonate are reached with dilute hydrochloric acid.
(i) Write the equations for the reaction.
(ii) What is the mass of 4.5 moles of calcium carbonate? (Relative molecular mass of calcium carbonate is 100).
(iii) What is the volume of carbon dioxide liberated at stp?
(iv) What mass of calcium chloride is formed? (Relative molecular mass of calcium chloride is 111).
(v) How many moles of HCl are used in this reaction? [5]
(b) The diagram shows an apparatus for the laboratory preparation of hydrogen chloride.
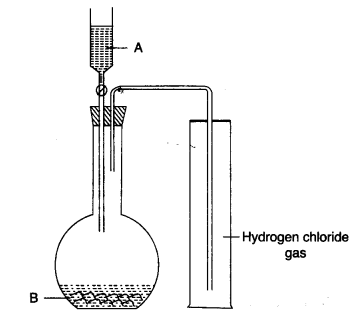
(i) Identify A and B.
(ii) Write the equation for the reaction.
(iii) How would you check whether or not the gas jar is filled with hydrogen chloride?
(iv) What does the method of collection tell you about the density of hydrogen chloride? [5]
Answer:
(a)
![]()

(b)
(i) A = Cone. Sulphuric acid.
B = Sodium chloride.
![]()
(iii) Bring a glass rod dipped in ammonium hydroxide near the mouth of the gas jar, it forms dense white fumes of ammonium chloride.
(iv) Collection of HCl ↑ by upward displacement of air proves that HCl ↑ is denser than air. It is 1-28 times heavier than air.
Question 5:
(a) Name the main constitutent metal in the following alloys:
- Duralumin.
- Brass
- Stainless steel. [3]
(b) An element has an atomic number 16. State:
- The period to which it belongs.
- The number of valence electrons.
- Whether it is a metal or non-metal. [3]
(c) Solution A is a sodium hydroxide solution. Solution B is a weak acid. Solution C is dilute sulphuric acid. Which solution will:
- liberate sulphur dioxide from sodium sulphite.
- give a white precipitate with zinc sulphate solution.
- contain solute molecules and ions? [3]
(d) By the addition of only one solution how would you distinguish between dilute hydrochloric acid and dilute nitric acid? [1]
Answer:
(a)
- Duralumin — Al, Mg, Mn, Cu.
- Brass — Cu, Zn
- Stainless steel — Fe, Cr, Ni, C
(b)
- Period = 3
- No. of valence electrons = 6
- It is a non metal.
(c)
- C—dil. sulphuric acid.
- A—Sodium hydroxide
- B—Weak acid.
(d) Add silver nitrate solution.
White ppt of silver chloride obtained with dil. hydrochloric acid.
No. ppt obtained with dil. nitric acid.
Question 6:
(a) Give the equation for the preparation of each of the following salts from the starting material given.
(i) Copper sulphate from copper(II) oxide.
(ii) Iron(III) chloride from Iron.
(iii) Potassium sulphate from potassium hydroxide solution.
(iv) Lead chloride from lead carbonate (two equations). [5]
(b) Compound A is bubbled through bromine dissolved in carbon tetrachloride and the product is CH2Br – CH2Br.
![]()
(i) Draw the structural formula of A.
(ii) What type of reaction has A undergone?
(iii) What is your observation?
(iv) Name (not formula) the compound formed when steam reacts with A in the presence of phosphoric acid.
(v) What is the procedure for converting the product of (b) (iv) back to A? [5]
Answer:
(a)
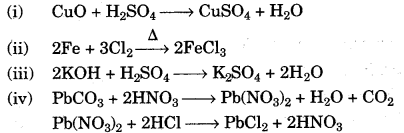
(b)
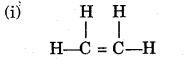
(ii) Addition reaction
(iii) Brown colour of bromine decolourized
(iv) Ethanol/Ethyl alcohol
(v) Dehydration.
Question 7:
(a) The diagram shows a simple arrangement of the fountain experiment:
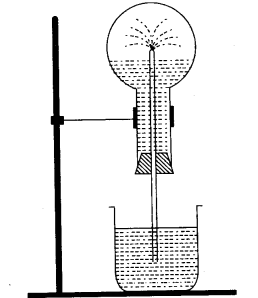
- Name the two gases you have studied which can he used in this experiment.
- What is the common property demonstrated by this experiment? [3]
(b) Define the followig terms:
- Ionization potential.
- Electron affinity. [2]
(c) The action of heat on the blue crystaline solid L gives a reddish brown gas M, a gas which re-lights a glowing splint and leaves a black residue. When gas N, which has a rotten egg smell, is passed through a solution of L a black precipitate is formed.
(i) Identify L, M and N (Name or formula)
(ii) Write the equation for the action of heat on L.
(iii) Write the equation for the reaction between the solution of L and the gas N. [5]
Answer:
(a)
- Ammonia gas and Hydrogen chloride gas.
- High solubility of the gases in water.
(b)
- Ionization potential is defined as the energy required to remove an electron from a neutral isolated gaseous atom to convert it into a positively charged gaseous ion.
- Electron affinity is the amount of energy released. While converting a neutral gaseous isolated atom into a negatively charged gaseous ion when an extra gaseous electron is added to the atom.
(c)
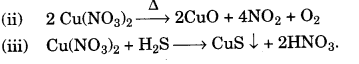
ICSE Class 10 Chemistry Previous Years Question Papers
