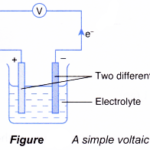
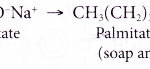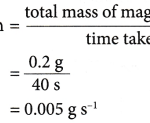Oxidation and Reduction in Chemical Cells In a simple voltaic cell, two different metals are in contact with an electrolyte.The more electropositive metal will become the negative terminal while the less electropositive metal will become the positive terminal.The chemical change that takes place at each electrode is actually a half-reaction … [Read more...] about Oxidation and Reduction in Chemical Cells
Explain the Cleansing Action Of Soaps and Detergents
Explain the Cleansing Action Of Soaps And Detergents The cleansing action of soaps and detergents:The cleansing action of both soaps and detergents results from their ability to lower the surface tension of water, to emulsify oil or grease and to hold them in a suspension in water. This ability is due to the structure of soaps and detergents. In water, a sodium … [Read more...] about Explain the Cleansing Action Of Soaps and Detergents
Redox Reactions by Transfer of Electrons at a Distance
Redox Reactions by Transfer of Electrons at a DistanceIn all redox reactions, electrons are transferred from the reducing agent to the oxidising agent. When the reducing and oxidising agents are mixed together as in the previous reactions, the transfer of electrons occurs quickly and cannot be detected. However, when the reducing and oxidising agents are separated … [Read more...] about Redox Reactions by Transfer of Electrons at a Distance
How do you calculate the reaction rate?
How do you calculate the reaction rate? Methods to measure the rate of reactionThe rate of reaction can be measured in two ways: (a) Average rate of reaction (b) Rate of reaction at a given timeThe average rate of reaction is the average value of the rate of reaction within a specified period of time. Example: 0.2 g of a magnesium ribbon reacts … [Read more...] about How do you calculate the reaction rate?
How is covalent bond is formed?
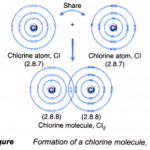
How is covalent bond is formed? A. Formation of a single covalent bonds Formation of chlorine molecule, Cl2A chlorine atom has an electron arrangement of 2.8.7. It has seven valence electrons. Each chlorine atom needs one more electron to achieve a stable octet electron arrangement (similar to argon). As a result, two chlorine atoms will combine with each … [Read more...] about How is covalent bond is formed?