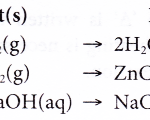Selina Concise Chemistry Class 10 ICSE Solutions Mole Concept and Stoichiometry APlusTopper.com provides step by step solutions for Selina Concise ICSE Solutions for Class 10 Chemistry Chapter 5 Mole Concept and Stoichiometry. You can download the Selina Concise Chemistry ICSE Solutions for Class 10 with Free PDF download option. Selina Publishers Concise Chemistry for Class … [Read more...] about Selina Concise Chemistry Class 10 ICSE Solutions Mole Concept and Stoichiometry
stoichiometry
How do you Write a Chemical Equation?
How do you Write a Chemical Equation? All chemical reactions are represented by chemical equations. A chemical equation is a shorthand representation of a chemical reaction using the symbols and formulae of substance involved in the chemical reaction. The symbols and formulae of the substances (elements or compounds) are arranged to show the reactants and products of a … [Read more...] about How do you Write a Chemical Equation?
What is stoichiometry and why is it used in chemistry?
What is stoichiometry and why is it used in chemistry?Stoichiometry is a study of quantitative composition of substances involved in the chemical reactions. A balanced chemical equation can be used to calculate number of moles, number of particles and mass or volume of a reactant or product. By using the information obtained from a balanced chemical equation, it is … [Read more...] about What is stoichiometry and why is it used in chemistry?