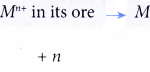Application of the reactivity series of metals in the extraction of metals Application of the reactivity series of metals towards oxygen in the extraction of metals:Only a few metals such as platinum, gold and silver are found free in nature. Other metals have to be extracted from their ores. Ores are naturally occurring rocks that contain high concentration … [Read more...] about Application of the reactivity series of metals in the extraction of metals
Rusting as a Redox Reaction
Rusting as a Redox Reaction
Rusting (Corrosion) as a Redox Reaction What is corrosion of a metal? Corrosion of metal:When metals are exposed to their environment, they undergo corrosion. For example, after some time, a shiny aluminium pot will lose its shine, silverware will tarnish and an iron structure will rust. Corrosion of metal is a redox reaction in which a metal is oxidised … [Read more...] about Rusting as a Redox Reaction
The Reactivity Series of Metals Towards Oxygen
The Reactivity Series of Metals Towards Oxygen The reactivity of metals differs from one metal to another. In fact, the form in which a metal occurs in nature depends on its reactivity. Gold has very low reactivity and therefore can be found in its metallic state in nature. Aluminium, potassium and sodium have very high reactivity, and therefore exist as … [Read more...] about The Reactivity Series of Metals Towards Oxygen