What is the collision theory in chemistry? According to the kinetic theory of matter, particles of matter are in continuous motion and constantly in collision with each other. For a reaction to occur, the particles of the reactants (atoms, molecules or ions) must touch each other through collision for bond breaking and bond formation to form the products. … [Read more...] about What is the collision theory in chemistry?
rate of reaction
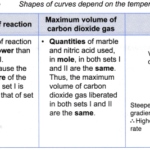
How does the temperature affect the rate of a chemical reaction?
How does the temperature affect the rate of a chemical reaction? Effect of temperature on the rate of reaction:When the temperature increases, the rate of reaction also increases. (a) For example, two sets of experiments are carried out using the reacting conditions below: Set I: 1 g of granulated zinc and 20 cm3 of 0.2 mol dm-3 hydrochloric acid at … [Read more...] about How does the temperature affect the rate of a chemical reaction?
How does the surface area affect the rate of reaction?
How does the size of particles affect the rate of reaction? Effect of surface area on the rate of reaction:When the particle size of a fixed mass of a solid reactant becomes smaller, the total exposed surface area becomes larger, the rate of reaction increases.For example, two sets of experiments are carried out using the reacting conditions below: Set I: 1 g of … [Read more...] about How does the surface area affect the rate of reaction?
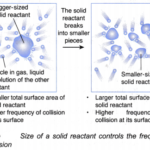
How does the collision theory affect the rate of reaction?
How does the collision theory affect the rate of reaction? Explaining the effect of size of a solid reactant/surface area on the rate of reaction using collision theoryWhen the size of a fixed mass of a solid reactant decreases, the rate of reaction increases. This can be explained using the collision theory, as below: (a) When the size of a fixed mass of a … [Read more...] about How does the collision theory affect the rate of reaction?