Plus Two Chemistry Chapter Wise Previous Questions Chapter 5 Surface Chemistry is part of Kerala Plus Two Chemistry Chapter Wise Previous Year Questions and Answers. Here we have given Plus Two Chemistry Chapter Wise Questions and Answers Chapter 5 Surface Chemistry.
Kerala Plus Two Chemistry Chapter Wise Previous Questions Chapter 5 Surface Chemistry
Question 1.
Colloids exhibit certain special properties. (March – 2010)
a) Name the property of colloid involved in the construction of ultramicroscope.
b) Explain the above property.
c) What are the conditions to be satisfied to exhibit Tyndall Effect?
Answer:
a) Tyndall effect
b) When a colloid is viewed at right angles to the passage of light, the path of the beam is illuminated by a bluish light. This effect is called the Tyndall effect and the bright cone of light observed is called the Tyndall cone. Tyndall effect is caused by the scattering of light by colloidal particles in all directions in space.
c) There are two conditions for observing the Tyndall effect:
- The diameter of the dispersed particles is not much smaller than the wavelength of the light used.
- The refractive indices .of the dispersed phase and the dispersion medium differ greatly in magnitude.
Question 1.
In an attempt to prepare ferric hydroxide sol by adding small amount of ferric chloride to water, one person got a precipitate of ferric hydroxide. (Say – 2010)
a) How can you help him to convert Fe(OH)3 precipitate to Fe(OH)3 sol?
b) Name the phenomenon behind this.
c) What happens when BaCI2 is added to Fe(OH)3 sol?
d) Give reason forthe above.
Answer:
a) By adding a solution of FeCI3 to the fresh precipitate of ferric hydroxide.
b) Peptization
c) Precipitation of Fe(OH)3 sol will take place.
d) BaCI2 being an electrolyte ionises to Ba2+ + and Cl ions. The particles of Fe(OH)3 precipitate adsorb Ba2+ ions to their surface and get positively charged. These positively charged particles of the precipitate repel each other and ultimately break up into smaller particles of the size of a colloid. Here BaCI2 is acting as a peptizing agent.
Question 1.
Physisorption and Chemisorption are 2 types of ad-sorption. (March – 2011)
a) What is the effect of temperature on physisorption and chemisorption?
b) In certain cases physisorption transfers into chemisorption as temperature is increased. Explain with an example.
c) Explain how colloids get coagulated on addition of salts.
Answer:
a) Physical adsorption or physisorption decreases with increase of temperature where as chemical adsorption increases with increase of temperature reaches a maximum value at an optimum temperature and then decreases with increase in temperature.
b) Physisorption of a gas adsorbed at low temperature may transform into chemisorption at a high temperature. This is due to the fact that no activation energy is required for physisorption while chemisorption requires activation energy. For example, dihydrogen is first adsorbed on nickel by van der Waals’ forces. This is physisorption. Molecules of dihydrogen then dissociate to form hydrogen atoms which are held on the surface by chemisorption.
c) When excess of a salt (electrolyte) is added, the colloidal particles interact with ions carrying charge opposite to that present on themselves. (According to Hardy-Schultz rule).
Question 1.
Ferric hydroxide sol can be prepared from freshly prepared ferric hydroxide precipitate. It can also be prepared by adding ferric chloride solution to boiling water. In both cases the sol particles are positively charged. (Say – 2011)
a) Name the above two methods of preparation of ferric hydroxide sol.
b) What happens when an electric potential is applied across two platinum electrodes dipping in ferric hydroxide sol? Explain.
Answer:
a) The method of preparation of ferric hydroxide sol from freshly prepared ferric hydroxide precipitate is called peptization. The method of preparation of ferric hydroxide sol by the addition of ferric chloride solution to boiling water is called hydrolysis,
b) When an electric potential is applied across two platinum electrodes dipped in ferric hydroxide sol electrophoresis will occur. Since ferric hydroxide sol is positively changed the colloidal particles will move towards the platinum electrode which acts as the cathode (negative electrode).
Question 1.
Colloids have many characteristic properties. Among this Tyndall effect is an optical property and coagulation is the process of settling of colloidal particles. (March – 2012)
i) What is the Tyndall effect?
ii) State Hardy Schulze rule which deals with the coagulation of colloids by the addition of an electrolyte.
iii) What is a protective colloid?
Answer:
i) When light passes through colloids the path of light becomes visible. This effect is called Tyndall effect. This is due to scattering of light by colloidal particles.
ii) It state that ‘Thegreaterthe valency of flocculating ion, the greater will be its coagulating, flocculating or precipitating power.
Note:
i) In the coagulation of a positive sol, the flocculating power increases in the order Na+ < Ba2+ + <AI3+
ii) Inthe coagulation of a negative sol, the flocculating power increases in the order Cl < S024 < PO34 < [Fe(CN)6]4 Na+ < Ba2+ < AI3+
iii) The lyophilic particles form a layer around the lyophobic particles and thus protect the latter from electrolytes. Such colloids are called protective colloids.
Eg : Gold sol can be protected by adding a little gelatin. Here, gelatin is the protective colloid.
Question 1.
Colloids are widely used in industry and in daily life. (Say – 2012)
i) What are colloids?
ii) Write any four applications of colloids.
Answer:
i) A colloid is a heterogeneous system in which one substance called a dispersed phase is dispersed as very fine particles in another substance called a dispersion medium. The particles in a colloid are larger than simple molecules but small enough to remain suspended. The diameter of colloidal particles ranges between 1nm and 1000 nm.
ii)
1) Electrical precipitation of smoke using Cottrell smoke precipitator-Smoke is a colloid of solid particles such as carbon, arsenic compounds, dust etc., in air. These particles are precipitated using high voltage electrodes.
2) Purification of drinking water – The suspended impurities present in water obtained from natural sources is coagulated by adding alum and is made fit for drinking purposes.
3) Medicines – Most of the medicines are colloidal in nature. Colloidal medicines are more effective because they have large surface area and are therefore easily assimilated.
4) In the rubber industry – Rubber latex is a colloid of negatively charged rubber particles which is coagulated to rubber by adding formic acid.
Question 1.
a) The accumulation of molecular species at the surface rather than in the bulk of a solid or liquid is termed adsorption. (March – 2013)
i) What is adsorption isotherm?
ii) Write the mathematical expression of Freundlich adsorption isotherm.
b) Enzymes are known as biochemical catalysts. Write any two important characteristics of enzyme catalysis.
Answer:
a) i) A plot between the amount of gas adsorbed pergram of adsorbent (x/m) and the pressure of the adsorbate at constant temperature is called Adsorption isotherm.
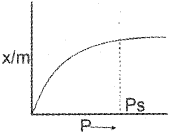
\(\frac{x}{m}=k p^{1 / n}\)
OR
\(\log \frac{x}{m}=\frac{1}{n} \log P+\log k[latex]
x → Amount of gas adsorbed by ‘m’ gram of the adsorbent at a pressure P. ‘k’ and ‘n’ are constants.
b)
- Enzymes are highly specific in nature.
- Enzymes are highly efficient.
- They are highly active under an optimum temperature.
Question 1.
There are mainly two types of adsorption of gases on solids (Say – 2013)
a) What are the two types of adsorption of gases on solids?
b) Write any two characteristics of each of the above two types of adsorption.
Answer:
a) Physical adsorption or physisorption and chemical adsorption or chemisorption,
b)
| Physical Adsorption | Chemical Adsorption |
| 1) Adsorbate molecules are held on the surface of the adsorbent by weak van der Waals’ forces | 1) The adsorbate molecules are held on the surface of the adsorbent by chemical bonds |
| 2) Not specific in nature | 2) Highly specific in nature |
| 3) Low temperature is favourable for adsorption | 3) High temperature is favourable for adsorption |
| 4) Enthalpy of adsorption is low (20 – 40 kJ mol-1) | 4) Enthalpy of adsorption is high (80 – 240kJ mol-1) |
Question 1.
Sols are colloidal systems in which dispersion medium is liquid and dispersed phase is solid. (March – 2014)
a) Write any four differences between lyophilic sols and lyophobic sols.
b) Peptisation is a method .of preparation of sols. Write a general procedure for peptisation.
Answer:
a)
| Lyophilic Sols | Lyophobic Sols |
| 1. Liquid-loving | 1. Liquid-hating |
| 2. Reversible | 2. Irreversible |
| 3. Stable | 3. Not stable |
| 4. Cannot be easily coagulated and no stabilising agent required | 4. Can be easily coagulated and need a stabilising agent for preservation |
b) Peptization is the process of converting a pre-cipitate into colloidal sol by shaking it with dispersion medium in the presence of small amount of electrolyte. The electrolyte used for this purpose is called peptizing agent. This method is applied to convert a freshly prepared precipitate into a colloidal sol. During peptization the pre- . cipitate adsorbs one of the ions of the electrolyte on its surface. This causes the development of positive or negative charge on precipitates, which ultimately break up into smaller particles of the size of a colloid.
Question 1.
a) ‘Adsorption’ has many applications. Write any two applications of adsorption. (Say – 2014)
b) Physisorption and chemisorption are the two types of adsorption. Write any four differences between them.
Answer:
a) 1) In production of high vacuum
2) In gas masks
b)
| Physical Adsorption | Chemical Adsorption |
| 1) Adsorbate molecules are held on the surface of the adsorbent by weak van der Waals’ forces | 1) The adsorbate molecules are held on the surface of the adsorbent by chemical bonds |
| 2) Not specific in nature | 2) Highly specific in nature |
| 3) Low temperature is favourable for adsorption | 3) High-temperature is favourable for adsorption |
| 4) Enthalpy of adsorption is low (20 – 40 kJ mol-1) | 4) Enthalpy of adsorption is high (80 – 240kJ mol-1) |
Question 1.
a) Which of the following is Lyophobic colloid? (March – 2015)
1) Starch in water
ii) Gum in water
iii) Soap in water
iv) Gold sol
b) Write four applications of colloids.
Answer:
a) iv) Gold sol
b)
- Electrical precipitation of smoke
- Purification of drinking water
- Medicines
- Tanning
- The cleansing action of soaps and detergents
- Photographic plates and films
- Coagulation of rubber latex etc. (Any four)
Question 1.
a) Which of the following is an example of absorption? (Say – 2015)
i) Water on silica gel
ii) Water on CaCI2
iii) Hydrogen on finely divided Nickel
iv) Oxygen on metal surface
b) Write any two differences between absorption and adsorption.
OR
Based on particles of dispersed phase, colloidal systems are classified into multimolecular, macro molecular and associated colloids.
a) Which of the following colloidal system is an example for multimolecular system?
i) Starch water
ii) Soap solution
iii) Ferric hydroxide in water
iv) Polyvinyl alcohol in water
b) Associated colloids are also known as micelles. How are they formed?
Answer:
a) ii) Water on CaCI2
b)
| Absorption | Adsorption |
| 1. It involves the uniform distribution of the molecular species throughout the bulk. | 1. It involves the unequal distribution of the molecular species in bulk and at the surface. |
| 2. It occurs throughout the body of material. | 2. It is a surface phenomenon. |
OR
a) iii) Ferric hydroxide in water.
b) Associated colloids or micelles are formed by the aggregation of ions of an electrolyte above a particular concentration and temperature. Soap is an example of associated colloid. It is sodium or potassium salt of higher fatty acids and can be represented as RCOO-Na+ or RCOO K+. When dissolved in water, it dissociates into RCOO and Na+ or K3 ions. The RCOO ions consist of two parts – a long hydrocarbon chain R (non-polar ‘tail’) which is hydrophobic (water-repelling), and a polar group COO (polar ionic ‘head’) whic is hydrophobic (water-loving). Therefore, the RCOO ions are present on the surface with their COO groups in water and the hydrocarbon chains (R) staying away from it and remain at the surface. At critical micelle concentraion, the anions are pulled into the bulk of the solution and aggregate to form a spherical shape with their hydrocarbon cains pointing towards the centre of the sphere with COO part remaining outward on the surface of the sphere. An aggregate thus formed is known as ionic micelle.
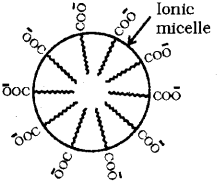
Question 1.
i) Catalysis can be classified into two groups homogenous and heterogeneous. (March – 2016)
a) What do you mean by homogenous catalysis?
b) Write one example for heterogeneous catalysis.
ii) Which of the following is an emulsifying agent?
a) Milk
b) Butter
c) Gum.
d) Lamp black
Answer:
i) a) When the reactants and the catalyst are in the same phase (i.e., liquid or gas), the process is said to be homogenous catalysis.
b) e.g. Oxidation of sulphur dioxide into sulphur trioxide in the presence of Pt or V2O5.
[latex]2 \mathrm{SO}_{2}(\mathrm{~g})+\mathrm{O}_{2}(\mathrm{~g}) \stackrel{\mathrm{Pt}(\mathrm{s})}{\longrightarrow} 2 \mathrm{SO}_{3}(\mathrm{~g})\)
In this process the reactants, sulphur dioxide and dioxygen are in the gaseous state while the catalyst Pt or V2O5 is in the solid state.
ii) c) gum (for O/W emulsions)
d) lamp black (for W/O emulsions)
Question 1.
Dispersed phase and dispersion medium are two phases of the colloidal system (Say – 2016)
a) Name the colloid in which dispersed phase is liquid and dispersion medium is solid.
i) Sol
ii) foam
iii) Emulsion
iv) Gel
b) Physisorption and Chemisorption are two types of adsorption. Write any four differences between them.
Answer:
a) iv) Gel
b) Refer March 2017 Question No 1 (a)
Question 1.
There are mainly two types of adsorption. They are physisorption and chemisorption. (March – 2017)
a) Differentiate between physisorption and chemisorption.
b) Write any two applications of adsorption.
Answer:
a) Ant two
| Physisorption | Chemisorption |
| 1) It is arises because of van der Waals force. | 1) It is caused by chemical bond formation. |
| 2) It is not specific in nature. | 2) It is highly specific in nature. |
| 3) It is reversible in nature. | 3) It is irreversible. |
| 4) More easily liquefiable gases are adsorbed readily. | 4) Gases which can react with the adsorbent show chemisorption. |
| 5) Enthalpy of adsorption is low (20-40 kJ mol-1) in this case. | 5) Enthalpy of adsorption is high (80-240 kJ mol-1) in this case. |
| 6) Low temperature is favourable for adsorption. It decreases with increase of temperature. | 6) Hig temperature is favourable for adsorption. It increases with the increase of temperature. |
| 7) No appreciable activation energy is needed. | 7) High activation energy is sometimes needed. |
| 8) It increases with an increase of surface area. | 8) It too increases with an increase of surface area. |
b) Production of high vacuum, gas masks, control of humidity, removal of colouring matter from solutions, heterogeneous catalysis, separation of inert gases, in curing diseases, Froth floatation process, adsorption indicators, chromatographic analysis etc. (Any two applications required).
Question 1.
a) Which among the following Is not an electrical property of colloids? (Say – 2017)
i) Electrophoresis
ii) Electro osmosis
iii) Coagulation
iv) Tyndal effect
b) Freundlich adsorption Isotherm is
x/m = a kp1/n where n > 1
Answer the following questions based on Freundlich adsorption isotherm:
i) What Is adsorption isotherm?
ii) Explain the terms In the above equation.
Answer:
iv) Tyndaleffed
b) i) lt is a curve obtained by plotting extent of adsorption against pressure at a constant temperature.
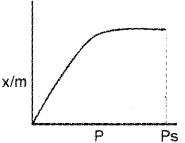
ii) x = mass of gas adsorbed
m = mass of adsorbent
p = pressure
k, n are constant n > 1
We hope the Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 5 Surface Chemistry help you. If you have any query regarding Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 5 Surface Chemistry, drop a comment below and we will get back to you at the earliest.
