Plus Two Chemistry Chapter Wise Previous Questions Chapter 15 Polymers is part of Kerala Plus Two Chemistry Chapter Wise Previous Year Questions and Answers. Here we have given Plus Two Chemistry Chapter Wise Questions and Answers Chapter 15 Polymers.
Kerala Plus Two Chemistry Chapter Wise Previous Questions Chapter 15 Polymers
Question 1.
Based on the mode of polymerisation we can classify polymers into addition polymers and condensation polymers. (March – 2010)
a) Classify the polymers given below into addition polymers and condensation polymers. Terylene, polyvinyl chloride, bakelite, polyethene.
b) How will you prepare Nylon 6,6?
Answer:
a) Addition polymers – Polyvinyl chloride, Polythene Condensation polymers-Terylene, Bakelite
b) Nylon 6,6 is prepared by the condensation ‘ polymerisation of hexamethylene diamine with adipic acid under high pressure and at high temperature.
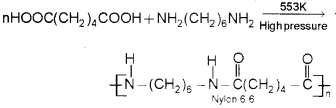
Question 1.
Polymers are high molecular mass compounds having special properties and so used for special purposes. Identify the following polymers X, Y and Z. (Say – 2010)
a) X is a polymer resistant to heat and chemicals. People used it to make non-sticky frying pans.
b) Y is a polymer formed from ethylene glycol and terephthalic acid and used for making heart valves.
c) Z is a polymer used for making unbreakable crockery items.
Answer:
a) Teflon (Polytetrafluoroethylene)
b) Terylene
c) Melamine – formaldehyde polymer
Question 1.
a) LDPE is a homopolymer, while Nylon 6,6 is a co-polymer. Explain. (March – 2011)
b) Classify the following into homopolymer or co-polymer: Nylon-6, HDPE.
Answer:
a) LDPE is Low-Density Poly Ethylene and its monomer is ethylene. The monomers of Nylon 6,6 are hexamethylene diamine and adipic acid. Here two monomers are present and hence it is a copolymer.
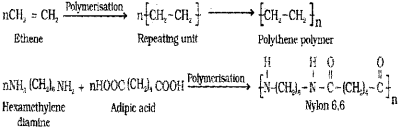
b) Nylon – 6 is a homopolymer contains only one type of monomer units, i.e., aminocaproic acid
Question 1.
Monomers polymerise to give polymers. Polymers can be classified in many ways. (Say – 2011)
a) Distinguish between homopolymers and co-polymers.
b) Give the name or formulae of the monomers in the following polymers.
i) Nylon – 6, 6
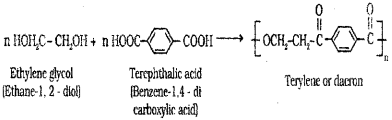
ii) Dacron
Answer:
a) Homopolymers-Addition polymers formed by the polymerisation of a single monomeric species.
e.g., Polythene.
Co-polymers – Polymers made by addition polymerisation from two different monomers.
e.g., Buna-S, Buna-N
b) i) Nylon 6,6 – hexamethylene diamine with adipic acid
ii) Dacron – Ethylene glycol (HOH2C-CH2OH) and Terephthalic acid( Hooc![]() COOH).
COOH).
Question 1.
a) Rubber is a natural polymer obtained from the bark of rubber trees. (March – 2012)
i) Name the monomer of natural rubber.
ii) Vulcanisation improves the elasticity of rubber. What is vulcanisation?
b) Write two examples for synthetic rubber.
Answer:
a) i) Isoprene, 3-butadiene)
ii) The process of heating a mixture of raw rubber with sulphur and an appropriate additive at a temperature range between 373 K to 415 K.
b) e.g. 1. Neoprene, 2. Buna – N
Question 1.
PVC, bakelite and polythene are plastics. (Say – 2012)
i) Classify the above plastics into thermoplastics and therm osetting plastics.
ii) Name the monomer units of PVC and bakelite.
Answer:
i) Thermo plastics – PVC, Polythene
Thermosetting plastics – Bakelite
ii) The monomer of PVC ¡s ployvinyl chloride (CH2=CH-Cl)
The monomers of bakelite are phenol (C6H5ÇOH) and formaldehyde (HCHO)
Question 1.
a) Synthetic rubber is a vulcanisable rubber-like polymer. (March – 2013)
1) Write one example for synthetic rubber.
ii) Write the method of preparation of the above synthetic rubber.
b) Which are the monomers of Nylon-6 and Nylon-66?
Answer:
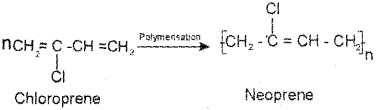
a) 1) Neoprene
2) It is obtained by the free radical polymerisation of chloroprene.
b) Polymer Monomer
1) Nylon 6 → Caprolactam
2) Nylon 6,6 → Hexamethylene diamine & Adipic acid
Question 1.
Natural rubber obtained from rubber latex is soft and sticky. (Say – 2013)
a) Suggest a method to improve the stiffness of rubber.
b) Explain the above method.
c) Classify the following into natural and synthetic polymers:
Nylon, Starch, Cellulose, PVC
Answer:
a) Vulcanisation
b) It is the process of heating a mixture of raw rub- ber with sulphur and an appropriate additive at a temperature range between 373 K to 415 K. On vulcanisation, sulphur forms cross-links at the reactive sites of double bonds in the polyisoprene chain and thus rubber gets stiffened. Vulcanisation improves the physical properties of natural rubber-like elasticity, water absorption capacity and solubility. It also increases its resistance to attack by oxidising agents.
c) Natural polymers – Cellulose, Starch Synthetic polymers – Nylon, PVC
Question 1.
a) Write any two differences between step-growth polymerisation and chain-growth polymerisation. (March – 2014)
b) What are the monomers of the following?
i) Neoprene
ii) Nylon-6
Answer:
a)
| Chain growth polymerisation | Step growth polymerisation |
| 1) Molecules of the same or different monomers add together on a large scale to form a polymer. | 1) There is repetitive condensation reaction between two bi-functional monomers with elimination of some simple molecules. |
| 2)The monomers are unsaturated compounds like alkenes, alkadienes and their derivatives. | 2)The monomers are saturated compounds with two functional groups. |
b) 1) Neoprene: Neoprene (2-Chloro-1 ,3-butadiene)

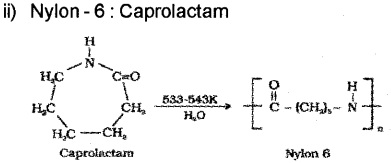
Question 1.
a) Name two thermoplastics. (Say – 2014)
b) Nylon 6, 6 and Dacron are two synthetic fibres. Suggest the monomers of each.
Answer:
a) 1) Polythene
2) Poly Vinyl Chloride (PVC)
b) Nylon 6,6: Hexamethylenediamine [HOOC(CH2)4 COOH] and adipic acid [H2N(CH2)6NH2],
Dacron : Ethylene glycol (HOH2C – CH2OH) and Terephthalic acid (HOOC – C6H4 – COOH).
Question 1.
Polymers are macromolecules formed by union of monomers. (March – 2015)
a) Name natural polymer and synthetic polymer.
b) Distinguish between thermoplastic and thermosetting polymers with example.
Answer:
a) Natural polymer – proteins, cellulose, starch, resins, natural rubber (any one)
Synthetic polymer- Polythene, Nylon 6,6, BunaS, Teflon, PVC (any one)
b) Thermoplastic polymers – These are linear or slightly branched long-chain molecules capable of repeatedly softening on heating and hardening on cooling. These polymers possess intermolecular forces of attraction intermediate between elastomers and fibres. These can be reused.
Example – polythene, polystyrene, polyvinyl (any one)
Thermosetting polymers – These polymers are cross linked or heavily branched molecules, which on heating undergo extensive cross-linking in moulds and again become infusible. These cannot be reused.
Example – bakelite, urea-formaldehyde resins (any one)
Question 1.
Polymers are classified into elastomers, fibres, thermoplastic and thermosetting plastics, depending upon the intermolecular forces. Fill in the vacant boxes given below: (Say – 2015)
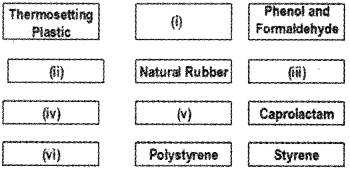
Answer:
- Bakelite
- Elastomer
- Isoprene (2-Methy-1,3-butadiene)
- Fibres
- Nylon 6
- Thermoplastic
Question 1.
Polymers can be classified based on molecular forces. (March – 2016)
a) Classify the following polymers into elastomers and fibres: Rubber, Nylon 6,6 Buna-S, Terylene
b) What do you mean by thermosetting polymers? Give one example.
Answer:
a) Elastomers – Rubber, Buna-S Fibres – Nylon 6,6, Terylene b) Thermosetting polymers are cross-linked or heavily branched molecules, which on heating undergo extensive cross-linking in moulds and again become infusible.
These cannot be reused.
e.g. Bakelite, urea-formaldehyde resins (any one)
Question 1.
Polymers are of different types (Say – 2016)
a) Identify the thermoplastic polymer from the following:
i) Bakelite
ii) Nylon-6,6
iii) Neoprene
iv) PVC
b) What is biodegradable polymers? Write an example.
Answer:
a) (iv) PVC
b) Biodegradable polymers – Polymers which can be degraded by microorganisms,
eg. Poly β-hydroxybutyrate – co-β-hydroxy- valerate (PHBV), Nylon 2-nylon 6 (any one example)
Question 1.
a) Which of the following is not applicable to Nylon 6,6? (March – 2017)
i) Synthetic polymer
ii) Fibre
iii) Addition polymer
iv) Condensation polymer.
b) Differentiate between thermoplastics and thermosetting plastics. Write one example each to them.
Answer:
a) Addition polymer
b)
| Thermoplastic | Thermosetting |
| 1. They can be reused | 1. Cannot be reused |
| 2. They can be repeatedly softening on heating and hardening on cooling | 2. On heating they undergo extensive cross-linking and become infusible |
| 3. They are linear or lightly branched long-chain polymers | 3. They are cross-linked or heavily branched polymers |
| 4. Eg. Polythene, Polystyrene, Polyvinyl | 4. Eg. Bakelite, Urea-formaldehyde, Resin. |
Question 1.
a) Distinguish between thermoplastic polymers and thermosetting polymers. (Say – 2017)
b) Name the monomers in the following two polymers.
i) Nylon 6,6
ii) Bu
Answer:
a) March 2017 Question 1 (b)
b) i) Nylon 66 → Adipic acid + Hexamethylene- diamine
ii) Buna-N → 1,3 butadiene + acrylonitrile
We hope the Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 15 Polymers help you. If you have any query regarding Kerala Plus Two Chemistry Chapter Wise Questions and Answers Chapter 15 Polymers, drop a comment below and we will get back to you at the earliest.
