Kerala Plus One Chemistry Notes Chapter 9 Hydrogen
Introduction
Hydrogen has the simplest atomic structure all the elements around us in nature. It consists of only one proton and one electron.
Position Of Hydrogen In The Periodic Table
Hydrogen is the first element in the periodic table. Hydrogen has electronic configuration 1 s1. On one hand, its electronic configuration is similar to the outer electronic configuration (ns¹) of alkali metals. On the other hand, it is short by one electron to the corresponding noble gas configuration, helium (1s²). It has resemblace to both alkali metals and halogens.
Dihydrogen, H2
Isotopes Of Hydrogen
There are three isotopes of hydrogen with mass numbers 1,2 and 3. They are called protium, deuterium and tritium respectively. Their natural abundances . are in the ratio l:1.56 × 10-2: 1 × 10-17 respectively.
- Protium (ordinary hydrogen)(\(_{ 1 }^{ 1 }{ H }\)): It is the most abundant isotope of hydrogen. Its nucleus contains one proton and no neutron.
- Deuterium (heavy hydrogen, \(_{ 1 }^{ 2 }{ H }\) or D): Heavy hydrogen is prepared from heavy water (D2O) which is obtained by electrolysis of ordinary water.
- Tritium has 2 neutrons in the nucleus.
Preparation Of Dihydrogen, H2
It is usually prepared by the following reactions:
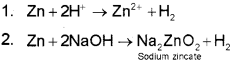
3. Reaction of steam on hydrocarbons or coke at high temperatures in the presence of catalyst yields hydrogen.
![]()
The mixture of CO and H2 is called water gas. As this mixture of CO and H2 is used for the synthesis of methanol and a number of hydrocarbons, it is also called synthesis gas or ‘syngas’. Nowadays ‘syngas’ is produced from sewage, sawdust, scrap wood, newspapers etc. The process of producing ‘syngas’ from coal is called ‘coal gasification’.
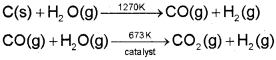
This reaction is called water-gas shift reaction.
Properties Of Dihydrogen
Physical Properties
Dihydrogen is a colourless, odourless, tasteless, combustible gas. It is lighter than air and insoluble in water.
Chemical Properties
Dihydrogen is not particularly reactive because of its high bond dissociation enthalpy. However, hydrogen forms compounds with almost all elements at high temperature or in presence of catalysts.
Reaction with halogens:
H2 (g) + X2(g) → 2HX(g) (X = F, Cl, Br, l)
Reaction with dioxygen:
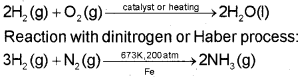
Uses Of Hydrogen
- Hydrogen is used in the manufacture of ammonia by Haber process, water gas, fertilisers etc.
- It is used in the hydrogenation of vegetable oils and as a reducing agent.
- It is used in the production of methanol and synthetic petrol.
- Liquid hydrogen is used in as rocket fuel along with liquid oxygen.
- It is used in oxy-hydrogen torch used for welding.
Hydrides
Hydrogen can form binary compounds with almost all elements. These are known as hydrides.
The hydrides are classified into three categories:
- Ionic or saline or salt like hydrides
- Covalent or molecular hydrides
- Metallic or non-stoichiometric hydrides
Ionic Or Saline Hydrides
These are stoichiometric compounds of dihydrogen formed with most of the s-block elements which are highly electropositive in character. However, significant covalent character is found in the lighter metal hydrides such as LiH, BeH2 and MgH2.
Covalent Or Molecular Hydride
Dihydrogen forms molecular compounds with most of the p-block elements. Most familiar examples are CH4, NH3, H2O and HF. For convenience hydrogen compounds of nonmetals have also been considered as hydrides. Molecular hydrides are further classified according to the relative numbers of electrons and bonds in their Lewis structure into :
- electron-deficient,
- electron-precise,and
- electron-rich hydrides.
Group13 elements form electron deficient compounds. They act as Lewis acids i.e., electron acceptors. eg.B2H6 Group 14 elements form electron precise compounds. They have required number of electrons. eg.CH4. Electron-rich hydrides have excess electrons which are present as lone pairs. Elements of group 15-17 form such compounds. (NH3 has 1 – lone pair, H2O – 2 and HF -3 lone pairs).They will behave as Lewis bases.
Metallic Or Non-Stoichiometric (Or Interstitial) Hydrides
These are formed by many d-block and f-block elements. However, the metals of group 7, 8 and 9 do not form hydride. Even from group 6, only chromium forms CrH. These hydrides conduct heat and electricity though not as efficiently as their parent metals do. Unlike saline hydrides, they are almost always nonstoichiometric, being deficient in hydrogen. For example, LaH2.87 & YbH2.55
Water
Water is a colourless tasteless liquid. A major part of all living organisms is made up of water.The unusual properties of water is due to the presence of extensive hydrogen bonding between water molecules.
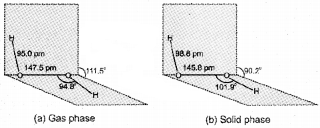
Structure Of Water
In the gas phase water is a bent molecule with a bond angle of 104.5°, and O-H bond length of 95.7 pm
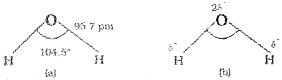
(a) The bent structure of water;
(b) the water molecule as a dipole
Structure Of Ice
The crystalline form of water is ice. At atmospheric pressure, ice crystallises in the hexagonal form, but at very low temperatures it condenses to cubic form. Hydrogen bonding gives ice a rather open type structure with wide holes. These holes can hold some other molecules of appropriate size interstitially. Density of ice is less than that of water. Therefore, an ice cube floats on water. In winter season ice formed on the surface of a lake provides thermal insulation which ensures the survival of the aquatic life.
Chemical Properties of Water
1) Amphoteric Nature:
It has the ability to act as an acid as well as a base i.e., it behaves as an amphoteric substance. In the Bronsted sense it acts as an acid with NH3 and a base with H2S.
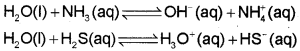
2) Redox Reactions Involving Water
Water can be reduced and oxidised:
2H2O(l) + 2Na(s) → 2NaOH(aq) + H2(g): reduction Water is oxidised to O2 during photosynthesis
6CO2(g) +12H2O(I) → C6H12O6 (aq) + 6H2O(I) + 6O2(g)
3) Hydrolysis Reaction:
Due to high dielectric constant, it has a very strong hydrating tendency.
P4O10(s) + 6H2O(l) → 4H3PO4(aq)
4) Hydrates Formation:
From aqueous solutions, many salts can be crystallised as hydrated salts. Such an association of water is of different types viz.,
i) Coordinated water e.g.,
[Cr(H2O)6]3+3Cl–
ii) Interstitial water.g., BaCl2.2H2O
iii) hydrogen-bonded water.g.,
[Cu(H2O)4]2+ SO42-.H2O in CuSO4.5H2O
Hard And Soft Water
Water which produces lather with soap solution readily is called soft water. For example, rainwater, distilled water etc. Water which does not produce lather with soap solution readily is called hard water, eg: Sea water, water from certain rivers.
Hardness of water is due to the presence of bicarbonates, chlorides and sulphates of calcium and magnesium. The calcium and magnesium ions present in hard water form insoluble salts with soap and prevent the formation of lather.
![]()
Temporary Hardness
Temporary hardness is due to the presence of mag-nesium and calcium hydrogencarbonates.
It can be removed by boiling.
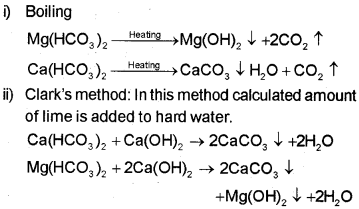
Permanent Hardness
It is due to the presence of soluble salts of magnesium and calcium in the form of chlorides and sulphates in water. Permanent hardness is not removed by boiling. It can be removed by the following methods:
i) Treatment with washing soda (sodium carbonate):
Washing soda reacts with soluble calcium and magnesium chlorides and sulphates in hard water to form insoluble carbonates.
MCl2 → MCO3 ↓ 2NaCl (M=Mg, Ca)
MSO4 + Na2CO3 → MCO3 ↓ +NaSO4
ii) Calgon’s method:
Sodium hexametaphosphate (Na6P6O18), commercially called ‘calgon’, when added to hard water, the following reactions take place.
Na6P6O18 → Na+ + Na4P6O182- (M=Mg, Ca)
M2+ + Na4P6O182- → [Na2MP6O18]2- + 2Na+
iii) Ion-exchange method:
This method is also called zeolite/perm utit process. Hydrated sodium aluminium silicate iszeolite/permutit.Forthe sake of simplicity, sodium aluminium silicate (NaAlSiO4) can be written as NaZ.
2NaZ(s) + M2+(aq) → MZ2(s) + 2Na+(aq) (M=Mg, Ca)
MZ2 (S) + 2NaCl(aq) → 2NaZ(s) + MCl2(aq)
iv) Synthetic resins method:
Nowadays hard . water is softened by using synthetic cation exchangers. This method is more efficient than zeolite process.Ion exchange resin (RSO3H) is changed to RNa by treating it with NaCI. Here R is resin anion.
2RNa(s) + M2+(aq) → R2M(s) + 2Na+(aq)
The resin exchanges Na+ ions with Ca2+ and Mg2+ ions present in hard waterto make the water soft.
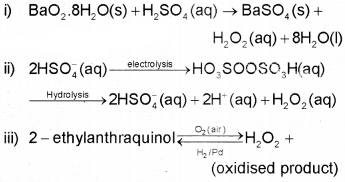
HYDROGEN PEROXIDE (H2O2)
It can be prepared by the following methods.
Structure
Hydrogen peroxide has a non-planar structure.
Chemical Properties
i) Oxidising action in acidic medium
PbS(s) + 4H2O2(aq) → PbSO4(s) + 4H2O(l)
ii) Reducing action in acidic medium
HOCl + H2O → H3O+ +Cl– + O2
iii) Oxidising action in basic medium
Mn2+ +H2O2 → Mn4+ + 2OH–
iv) Reducing action in basic medium
2MnO4– + 3H2O2 → MnO2 + O2 + 2H2O + OH–
Uses
- As a bleaching agent for textiles, wood and paper pulp
- In the manufacture of chemicals such as sodium perborate, epoxides etc.
- A dilute solution of H2O2 is used as a disinfectant. This solution is used as an antiseptic for wounds, teeth and ears under the name perhydrol.
- iv) It is used in pollution control treatment of domestic and industrial effluents.
Heavy water. D2O
It is extensively used as a moderator in nuclear reactors and in exchange reactions for the study of reaction mechanisms. It can be prepared by exhaustive electrolysis of water or as a by-product in some fertilizer industries.lt is used for the preparation of other deuterium compounds.
Dihydrogen As A Fuel
Due to extensive use, our reserves of fossil fuels are fast depleting. A prospective alternative in this regard is what is known as hydrogen economy. The major idea behind hydrogen economy is the storage and transportation of energy in the form of gaseous and liquid hydrogen. Hydrogen can replace fossil fuels in automobiles, and coal or coke in industrial processes involving reduction. Hydrogen fuel can release more energy per unit weight of the fuel than our conventional fuels. Hydrogen oxygen fuel cells can be used for generating power in automobiles. Liquid hydrogen has already been used as rocket fuel along with liquid oxygen.
The technology involves the production of bulk quantities of hydrogen and its storage in liquid form in vacuum insulated cryogenic tanks. Transport of liquid hydrogen by road or rail, or through pipelines is feasible. Certain metal alloys can be used as smaller storage units for hydrogen.
