Kerala Plus One Chemistry Notes Chapter 10 The s Block Elements
Introduction
Group 1 of the periodic table consists of the elements: Lithium, Sodium, Potassium, Rubidium, Caesium and Francium. They are collectively known as alkali metals.
Group 2 consists of Beryllium, Mgnesium, Calcium, Strontium, Barium and Radium. These elements except of beryllium are known as the alkaline earth metals. The general electronic configuration of s-block elements is [noble gasjns1 for alkali metals and [noble gas] ns² for alkaline earth metals. The first elements of Group 1 and Group 2 respectively exhibit diagonal similarity, which is commonly referred to as diagonal relationship in the periodic table. The diagonal relationship is due to the similarity in ionic sizes and /or charge/radius ratio of the elements.
Group 1 Elements: Alkali Metals
1) Electronic Configuration:
All the alkali metals have one valence electron, ns¹ outside the noble gas core. The loosely held s-electron readily lose electron to give monovalent M+ ions.
2) Atomic And Ionic Radii:
The atomic and ionic radii of alkali metals increase on moving down the group. Hence, ionization enthalpies of the alkali metals are considerably low and decrease down the group.
3) Hydration Enthalpy:
The hydration enthalpies of alkali metal ions decrease with increase in ionic sizes. Li+ > Na+ > K+ > Rb+ > Cs+ Li+ has maximum degree of hydration and for this reason lithium salts are mostly hydrated, e.g., LiCl- 2H2O
Physical Properties
When heat is supplied to alkali metal or its salt the electrons are excited to higher energy levels. As these electrons return to their original level; radiations are emitted which fall in the visible region of electromagnetic spectrum. Thus they appear coloured. Li imparts crimson red colour, K gives violet colour and Na gives golden yellow colour to the flame.
Chemical Properties
The reactivity of these metals increases with their size. They burn vigorously in oxygen forming oxides. Lithium forms monoxide, sodium forms peroxide, the other metals form superoxides. The superoxide O2 ion is stable only in the presence of large cations such as K, Rb, Cs.
4Li + O2 → 2LizO(oxide)
2Na + O2 → Na2O2 (peroxide)
M + O2 → MO2(superoxide)
(M=K, Rb, Cs)
Because of their high reactivity towards air and water, they are normally kept in kerosene oil.lt may be noted that although lithium has most negative E° value.
![]()
They also react with proton donors such as alcohol, gaseous ammonia and alkynes.AII the alkali metal hydrides are ionic solids with high melting points.
2M + H2 → 2M+H–.
The alkali metals readily react vigorously with halogens to form ionic halides, M+X–. However, lithium halides are somewhat covalent. It is because of the high polarisation capability of lithium-ion. The alkali metals are strong reducing agents, lithium being the most and sodium the least powerful. The alkali metals dissolve in liquid ammonia giving deep blue solutions. The solutions are paramagnetic and on standing slowly liberate hydrogen.
General Characteristics Of The Compounds Of The Alkali Metals
Oxides And Hydroxides
Reactivity of alkali metals with oxygen increases down the group. Lithium, when heated in air, forms the normal oxide (Li2O) while sodium forms the per-oxide (Na2O2). Potassium, Rubidium and caesium form superoxides (MO2).
4Li + O2 → 2Li2O; 2Na+ O2 → Na2O2; K + O2 → KO2
The normal oxides dissolve in water to form hydroxides (MOH) which are strong bases. However, LiOH is only slightly soluble in water and it decomposes on heating. The peroxides and superoxides also dis-solve in water to form basic hydroxides. The basic character of alkali metal hydroxides increases down the group.
Halides
Alkali metals react vigorously with halogens to form metal halides of the general formula MX. 2M+X2 → 2MX X=F, Cl, Br or l and M= alkali metal Reactivity of alkali metal towards halogen increases from Li to Cs. Halides of alkali metals are ionic compounds readily soluble in water. But LiF is almost insoluble due to high lattice energy.
Anomalous Properties Of Lithium
The anomalous behaviour of lithium is due to the :
- exceptionally small size of its atom and ion, and
- high polarising power (i.e., charge/ radius ratio).
As a result, there is increased covalent character of lithium compounds which is responsible for their solubility in organic solvents.
Points Of Similarities Between Lithium And Magnesium
The similarity between lithium and magnesium is particularly striking and arises because of their similar sizes: atomic radii, Li = 152 pm, Mg= 160 pm; ionic radii: Li+ = 76 pm, Mg2+ = 72 pm. The main points of similarity are:
- Both lithium and magnesium are hander and lighter than other elements in the respective groups.
- Lithium and magnesium react slowly with water. Their oxides and hydroxides are much less soluble and their hydroxides decompose on heating. Both form a nitride, Li3N and Mg3N2, by direct combination with nitrogen.
- The oxides, Li2O and MgO do not combine with excess oxygen to give any superoxide.
- The carbonates of lithium and magnesium decompose easily on heating to form the oxides and CO2.
Some Important Compounds Of Sodium Sodium Carbonate (Washing Soda), Na2CO3.10H2O
Sodium carbonate is generally prepared by Solvay Process.
The equations for the complete process may be written as:
2NH3 + H2O + CO2 → (NH4)2CO3
(NH4)2CO3 + H2O + CO2 → 2NH4HCO3
NH4HCO3 +NaCl → NH4Cl + NaHCO3
2NaHCO3 → Na2CO3 +CO2 +H2O
In this process, NH3 is recovered when the solution containing NH4Cl is treated with Ca(OH)2. On heating washing soda becomes monohydrate and then completely anhydrous i.e., soda ash.
Sodium Chloride, NaCl
The most abundant source of sodium chloride is seawater. Common salt is generally obtained by evaporation of seawater. Crude sodium chloride, generally obtained by crystallisation of brine solution, contains sodium sulphate, calcium sulphate, calcium chloride and magnesium chloride as impurities. Calcium chloride, CaCl2, and magnesium chloride, MgCl2 are impurities because they are deliquescent (absorb moisture easily from the atmosphere). To obtain pure sodium chloride, the crude salt is dissolved in minimum amount of water and filtered to remove insoluble impurities. The solution is then saturated with hydrogen chloride gas. Crystals of pure sodium chloride separate out. Calcium and magnesium chloride, being more soluble than sodium chloride, remain in solution.
Uses:
- It is used as a common salt or table salt for domestic purpose.
- It is used for the preparation of Na2O2, Na0H and Na2CO3.
Sodium Hydroxide (Caustic Soda), NaOH
Sodium hydroxide is generally prepared commercially by the electrolysis of sodium chloride in Castner-Kellner cell. A brine solution is electrolysed using a mercury cathode and a carbon anode.
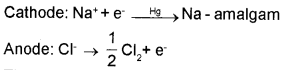
The amalgam is treated with water to give sodium hydroxide and hydrogen gas.
2 Na – amalgam + 2H2O → 2NaOH + 2Hg +H2
The sodium hydroxide solution at the surface reacts with the C02 in the atmosphere to form Na2CO3.
Uses:
It is used in (i)the manufacture of soap, paper, artificial silk and a number of chemicals,(ii) in petroleum refining, (iii) in the purification of bauxite, (iv) in the textile industries for mercerising cotton fabrics and (v) for the preparation of pure fats and oils.
Biological Importance Of Sodium And Potassium
Sodium ions participate in the transmission of nerve signals. The concentration gradient of Na+ and K+ demonstrates that-a discriminatory mechanism called sodium-potassium pump, operates across the cell membranes.
Group 2 Elements: Alkaline Earth Metals
The group 2 elements (except beryllium) are known as alkaline earth metals. The first element beryllium differs from the rest of the members and shows diagonal relationship to aluminium.
1) Electronic Configuration:
These elements have two electrons in the s-orbital of the valence shell. Their general electronic configuration may be represented as [noble gas] ns².
2) Atomic And Ionic Radii:
Within the group, the atomic and ionic radii increase with increase in atomic number due to the increased nuclear charge in these elements. They have low ionisation enthalpy and it decreases down the group with increase in size.
3) Hydration Enthalpy:
Hydration enthalpies of alkaline earth metal ions decrease with increase in ionic size down the group. Be2+ > Mg2+ > Ca2+ > Sr2+ > Ba2+ The hydration enthalpies of alkaline earth metal ions are larger than those of alkali metal ions.
Physical Properties
Calcium, Strontium and Barium impart characteristic brick red, crimson and apple green colours respectively to the flame. Inflame the electrons are excited to higher energy levels and when they drop back to the ground state, energy is emitted in the form of visible light. The electrons in Be and Mg are too strongly bound to get excited by flame. Hence, these elements do not impart any colour to the flame.
Chemical Properties
The alkaline earth metals are less reactive than the alkali metals. The reactivity of these elements increases on going down the group.
Reactivity towards air and water Beryllium and Magnesium are kinetically inert to oxygen and water because of the formation of an oxide film on their surface. However, powdered beryllium burns brilliantly on ignition in air to give BeO and Be3N2.
Reactivity towards halogen
M + X2 → MX2 (X = F, Cl, Br, I)
Reactivity towards hydrogen
All the elements except beryllium form their hydrides, MH2.BeH2, however, can be prepared by the reaction of BeCl2 with LiAlH4.
2BeCl2 +LiAlH4 → 2BeH2 +LiCl + AlCl3
Reactivity towards acids:
The alkaline earth metals readily react with acids liberating dihydrogen.
General Characteristics Of Compounds Of The Alkaline Earth Metals
i) Oxides and Hydroxides: Alkaline earth metals burn in air or oxygen to form their oxides. (Oxides are also prepared by the thermal decomposition of their carbonates). Be, Mg and Ca form monoxides (MO). The tendency to form peroxide increases as the size of the metal ion increases. Strontium and barium form peroxides (MO2)
2M + O2 → MO (M = Be, Mg or Ca)
M+O2 → MO2 (M = Sr or Ba)
BeO is amphoteric in character, while the oxides of the rest of the elements in group 2 are basic. The oxides of Ca, Sr and Ba react with water to form their corresponding hydroxides.
The hydroxides of alkaline earth metals are bases except Li(OH)2 which is amphoteric. The basic strength increases from Mg(OH)2 to Ba(OH)2. The solubility and thermal stability of hydroxides increase downward in the group. Be(OH)2 and Mg(OH)2 are almost insoluble. Ca(OH)2 is sparingly soluble, while Sr(OH)2 and Ba(OH)2 are increasingly more soluble.
ii) Halides: Group 2 metals directly combine with halogen to form divalent halides of the formula
The s-Block Elements
MX2 where X is the halogen. The metal halides are also formed by the action of halogen acids on metals, their oxides, carbonates and hydroxides. BeCl2 is, however, prepared by passing Cl2 over a hot mixture of BeO and coke.
In contrast to the halides of other alkaline earth metals, beryllium halides are covalent. In the solid-state BeCl2 has a polymeric chain structure involving Be-CI-Be bridges. The anhydrous halides are hygroscopic and form hydrates such as MgCl2.6H2O, CaCl2.6H2O etc. Due to this reason, anhydrous calcium chloride is widely used as a dehydrating agent. Fluorides are relatively less soluble due to high lattice energies,
iii) Salts of Oxoacids:
The alkaline earth metals also form salts of oxoacids. Some of these are : Carbonates, Sulphates and Nitrates.
Anomalous Behaviour Of Beryllium
Beryllium differs from the rest element in many of its properties. These are
- Beryllium has high ionisation enthalpy.
- Small size of Be atom
- Be does not exhibit coordination number more than four.
- The oxides and hydroxides of Be are amphoteric in nature.
Diagonal Relationship Between Beryllium And Aluminium
The ionic radius of Be2+ is estimated to be same as that of the Al3+ ion. Hence Be resembles Al in some ways. Some of the similarities are:
- Like AI, Be is not readily attacked by acids because of the presence of an oxide film on the surface of the metal.
- Beryllium hydroxide dissolves in excess of alkali to give a beryllate ion just as aluminium hydroxide gives aluminate ion.
- The chlorides of both Be and Al have Ch bridged chloride structure in vapour phase. Both the chlorides are soluble in organic solvents and are strong Lewis acids. They are used as Friedel Craft catalysts.
- Be and Al ions have strong tendency to form complexes, BeF42-, AlF63-.
Some Important Compounds Of Calcium
Important compounds of calcium and their preparations are given below.
Calcium Oxide Or Quick Lime, CaO
It is prepared by the following reaction.
CaCO3 \(\rightleftharpoons \) Ca0 + CO2
CO2 is removed as soon as it is produced to enable the reaction to proceed to completion.
CaO + H2O → Ca(OH)2
This process is called slaking of lime. CaO is a basis oxide.
Uses:
- Primary material for manufacturing cement
- It is used in the manufacturing of caustic soda
- Used to purify sugar
Calcium Hydroxide (Slaked Lime), Ca(OH)2
It is prepared by adding water to CaO. The aqueous solution of Ca(OH)2 is known as lime water and the suspension of slaked lime is known as milk of lime. When CO2 is passed through lime water it turns milky due to the formation of CaCO3
Ca(OH)2 + CO2 → CaCO3 +H2O
Uses:
- It is used in whitewash due to its disinfectant nature.
- Used in the preparation of bleaching powder.
- Used to purify sugar.
Calcium Carbonate, CaCO2
It occurs in limestone, chalk, marble etc.
It can be prepared by the following reactions.
Ca(OH)2 + CO2 → CaCO3 + H2O
CaCl2 + Na2CO3 → CaCO3 + 2NaCl
CaCO3 reacts with dilute acids to liberate carbon dioxide.
Uses:
- It is used as a flux in the extraction of metals.
- It is used as the building material of quick lime.
Calcium Sulphate (Plaster Of Paris), CaSO4.½H2O
It is obtained by heating gypsum (CaSO2.2H2O)
![]()
Above 393K anhydrous calcium sulphate is formed. This is known as ‘dead burnt plaster’
Used:
- It is used in building industry as well as plasters.
- Used to make casts of statues.
Cement
Cement is prepared by combining CaO with other materials such as clay with silica, SiO2 along with Oxides of Al, iron and magnesium. The average composition of portland cement is:
CaO, 50-60%;
SiO2, 20-25%;
Al2O3, 5-10%;
MgO, 2-3%;
Fe2O3, 1-2% and
SO3, 1-2%.
When limestone and clay are heated we get cement clinker. This clinker is mixed with gypsum to form cement.
Setting of Cement:
When mixed with water the setting of cement takes place to give a hard mass. It is due to the rearrangement and hydration of molecules of constituents. Gypsum is added to slow down the setting process so it gets sufficiently hardened.
Uses:
- Used in construction of building.
Biological Importance Of Magnesium And Calcium
Human body contains about 25g of Mg and 1200g of Ca. Mg is a cofactor in enzymes which use ATP in phosphate transfer process in our body. Photosynthesis in plants takes place in presence of chlorophyll which contains Mg. About 99% of body calcium is found in teeth and bones. Calcium concentration in plasma is regulated at 100mg/litre in presence of hormones such as calcitonin and parathyroid hormone.
