ISC Chemistry Previous Year Question Paper 2015 Solved for Class 12
Maximum Marks: 70
Time allowed: 3 hours
- Answer all questions in Part I and six questions from Part II, choosing two questions from Section A, two from Section B and two from Section C.
- All working, including rough work, should be done on the same sheet as, and adjacent to, the rest of the answer.
- The intended marks for questions or parts of questions are given in brackets [ ].
- Balanced equations must be given wherever possible and diagrams where they are helpful.
- When solving numerical problems, all essential working must be shown.
- In working out problems use the following data:
Gas constant R = 1.987 cal deg-1 mol-1 = 8.314 JK-1 mol-1 = 0.0821 dm3 atm K-1 mol-1. 1L atm = 1 dm3 atm = 101.3 J.
1 Faraday = 96500 Coulombs.
Avogadro’s number = 6.023 × 1023
Part – I
(Answer all questions)
Question 1.
(a) Fill in the blanks by choosing the appropriate word/words from those given in the brackets: [5]
(increases, decreases, positive, efficient, 68, non-efficient, no α-hydrogen, α-hydrogen, negative, Rosenmund’s, greater, Cannizzaro, 74, common-ion effect, lesser, buffer action, diamagnetic, paramagnetic)
(i) The more ……………. the standard reduction potential of metal, the …………. is its ability to displace hydrogen from acids.
(ii) Both ccp and hep are …………. packings and occupy about ………….. % of the available space.
(iii) The solubility of silver chloride ………….. in the presence of sodium chloride because of …………
(iv) Benzaldehyde undergoes …………… reaction on treatment with concentrated sodium hydroxide because it has ………. atom.
(v) The transition metals show ………….. character because of the presence of impaired electrons and Cu+ is ……….. because its electronic configuration is [Ar] 3d10.
(b) Complete the following statements by selecting the correct alternative from the choices given: [5]
(i) The molal freezing point constant of water is 1.86 K kg mol-1. Therefore, the freezing point of 0.1 M NaCl solution in water is expected to be:
(1) -1.86°C
(2) -0.372°C
(3) -0.186°C
(4) +0.372°C
(ii) For a first-order reaction, the rate constant for the decomposition of N2O5 is 6 × 10-4 sec-1. The half-life period for the decomposition in seconds is:
(1) 11.55
(2) 115.5
(3) 1155
(4) 1.155
(iii) When acetaldehyde is treated with Grignard reagent, followed by hydrolysis the product formed is:
(1) Primary alcohol
(2) Secondary alcohol
(3) Carboxylic acid
(4) Tertiary alcohol
(iv) The geometry of XeF6 molecule and the hybridization of the Xe atom in the molecule is:
(1) Distorted octahedral and sp3d3
(2) Square planar and sp3d2
(3) Pyramidal and sp3
(4) Octahedral and sp3d3
(v) In the complexes [Fe(CN)6]3- and [Pt(en) (H2O)2 (NO2) (Cl)]2+ the respective oxidation numbers of central metal atoms are:
(1) +3 and+4
(2) +6 and+4
(3) +6 and +3
(4) +3 and +3
(c) Answer the following questions: [5]
(i) What is the effect of temperature on the ionic product of water? How will it change the pH value of a neutral solution?
(ii) How many hours does it take to reduce 3 moles of Fe3+ to Fe2+ with 2.0 A current intensity?
(iii) How is urea prepared by Wohler synthesis?
(iv) Two liquids A and B form type II non-ideal solution which shows a minimum in its temperature mole fraction plot (T- χ diagram). Can the two liquids be completely separated by fractional distillation?
(v) The aqueous solution of sodium acetate is basic. Explain.
(d) Match the following: [5]
| (i) Disaccharide | (a) Lucas reagent |
| (ii) Carbylamine | (b) Condensation polymer |
| (iii) Dacron | (c) Obnoxious smell |
| (iv) Low spin complex, d2sp3 | (d) Sucrose |
| (v) Anhydrous ZnCl2 + conc. HCl | (e) Hexaamminecobalt (III) ion |
Answer:
(a) (i) common ion effect, lesser
(ii) non-efficient, 68
(ii) decreases, buffer action
(iv) Cannizzaro, α-hydrogen
(v) paramagnetic, diamagnetic.
(b) (i) -3
(ii) -4
(iii) -3
(iv) -3
(v) -2
(c) (i) \(\mathrm{H}_{2} \mathrm{O} \rightleftharpoons \mathrm{H}^{+}(a q)+\mathrm{OH}^{-}(a q)\) …(i)
At 25°C, [H+] = [OH–] = 1.01 × 10-7 M.
Kw = 10-14, K = 1.8 × 10-6 (at 25°C)
The value of Kw increases with the increase of temperature i.e., the concentration of H+ and OH– ions increases with increase in temperature. Neutral solution pH will increase.
(ii) Fe3+ + e– → Fe2+
3 mole Fe3+ will require 3-mole electrons.
Required charge = 3 × 96500 coulomb = 289500 coulomb
Charge = Current × Time
\(\text { Time }=\frac{\text { Charge }}{\text { Current }}=\frac{289500}{2}=1.44750 \times 10^{5} \mathrm{sec} .=40 \mathrm{hrs}\)
(iii) By Vital force theory
NH2CONH2 → CO(NH2)2
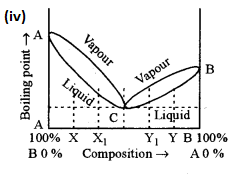
(v) CH3COONa → CH+3 + COONa–
Aqueous solution of CH3COONa is basic in nature.
(d) (i) (d)
(ii) (a)
(iii) (b)
(iv) (e)
(v) (c)
Part – II
Answer six questions choosing two from Section A, two from Section B and two from Section C.
Section – A
(Answer any two questions)
Question 2.
(a) (i) A solution containing 0.5 g of KCl dissolves in 100 g of water and freezes at -0.24°C. Calculate the degree of dissociation of the salt. (Kf for water = 1.86°C), Atomic weights [K = 39, Cl = 35.5] [3]
(ii) If 1.71 g of sugar (molar mass = 342) are dissolved in 500 mL of an aqueous solution at 300 K, what will be its osmotic pressure? [1]
(iii) 0.70 g of an organic compound, when dissolved in 32 g of acetone produces an elevation of 0.25°C in the boiling point. Calculate the molecular mass of organic compound (Kb for acetone = 1.72 K kg mol-1) [1]
(b) (i) What is the difference between the order of a reaction and the molecularity of a reaction? [2]
(ii) A substance decomposes by following first-order kinetics. If 50% of the compound is decomposed in 120 minutes, how long will it take for 90% of the compound to decompose? [2]
(c) Name the crystal structure of the copper metal. [1]
Answer:
(a) (i) w = 0.5 g of KCl
Kf = 1.86°C
W = 100 g of water
m of KCl = K + Cl = 39 + 35.5 = 74.5 g


Question 3.
(a) (i) Chromium metal crystallises with a body-centred cubic lattice. The edge length of the unit cell is found to be 287 pm. Calculate the atomic radius. What would be the density of chromium in g/cm3? (atomic mass of Cr = 52.99) [2]
(ii) Why does sodium chloride on heating with sodium vapours acquire yellow colour? [1]
(iii) The equilibrium constant for the reaction:
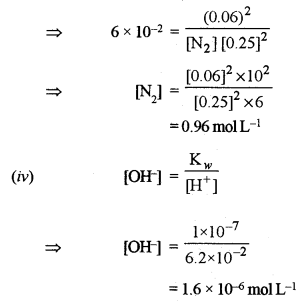
\(\mathrm{N}_{2}(g)+3 \mathrm{H}_{2}(g) \rightleftharpoons 2 \mathrm{NH}_{3}(g) \text { at } 715 \mathrm{K} \text { , is } 6.0 \times 10^{-2}\)
If, in a particular reaction, there are 0.25 mol L-1 of H2 and 0.06 mol L-1 of NH3 present, calculate the concentration of N2 at equilibrium.
(iv) Calculate the concentration of OH– ions in solution when [H+] = 6.2 × 10-2 mol L-1. [1]
(v) State Le Chatelier’s principle. [1]
(b) For a crystal of sodium chloride, state: [2]
(i) The type of lattice in which it crystallises.
(ii) The coordination number of each sodium ion and chloride ion in the crystal lattice.
(iii) The number of sodium ions and chloride ions present in a unit cell of sodium chloride.
(iv) The structural arrangement of the sodium chloride crystal.
(c) Consider the following reaction: [2]
\(\mathrm{N}_{2} \mathrm{O}_{4}(g)+\text { Heat } \rightleftharpoons 2 \mathrm{NO}_{2}(g)\)
How is the composition of the equilibrium mixture affected by:
(i) a change in temperature
(ii) a change in pressure
(iii) a change in concentration of N2O4
(iv) the removal of NO2 from the reaction mixture?
Answer:

(v) When a chemical system at equilibrium is subjected to a change in concentration, pressure or temperature; the equilibrium shifts in the direction of that counteracts the change.
(b) (i) Cubic close-packing (ccp) or face-centred cubic (fee) arrangement.
(ii) The coordination of each Na+ ion as well as of each Cl– ion is 6 and the structure possesses 6 : 6 coordination.
(iii) The unit cell of sodium chloride, eight Cl– ions are present at the comers and six Cl– ions at the face-centres.
Number of Cl– ions = \(8 \times \frac{1}{8}+6 \times \frac{1}{2}=4\)
Number of Na+ ions = \(12 \times \frac{1}{4}+1 \times 1=4\)
(iv) Rock salt structure.
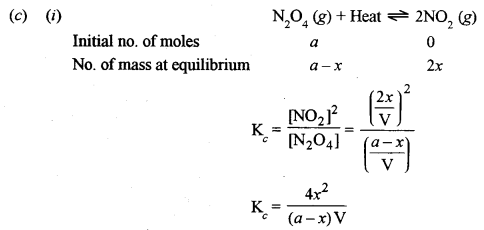
(ii) An increase in pressure would lead to a decrease in the dissociation of N2O4.
(iii) If the concentration of N2O4 is increased, the value of \(\mathrm{K}_{c}=\frac{4 x^{2}}{(a-x) \mathrm{V}}\)
increases. If the concentration of NO2 is increased, the value of K in expression remains constant.
(iv) Some NO2 get converted into N2O4. Hence, an increase in the concentration of NO2 will suppress the dissociation of N2O4.
Question 4.
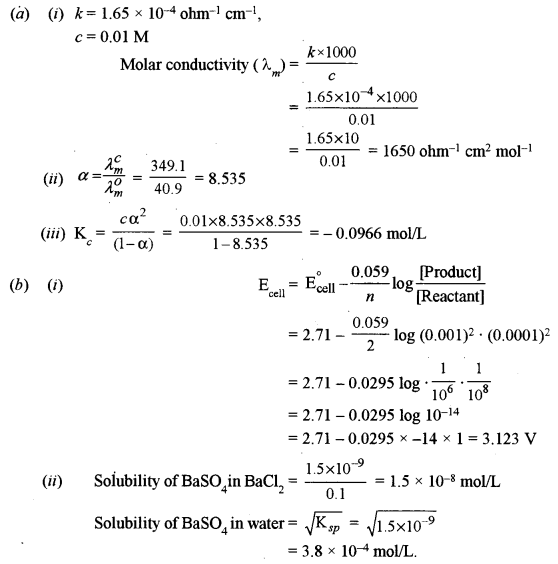
(a) The specific conductance of a 0.01 M solution of acetic acid at 298 K is 1.65 × 10-4 ohm-1. The molar conductance at infinite dilution for H+ ion and CH3COO– ion is 349.1 ohm-1 cm2 mol-1 and 40.9 ohm-1 cm2 mol-1 respectively. Calculate:
(i) Molar conductance of the solution.
(ii) Degree of dissociation of CH3COOH.
(iii) Dissociation constant of acetic acid.
(b) (i) Calculate the e.m.f. of the following cell reaction at 298 K:
Mg(s) + Cu2+ (0.0001 M) → Mg2+ (0.001M) + Cu(s)
The standard potential (E0) of the cell is 2.71 V.
(ii) The solubility product (Ksp) of BaSO4 is 1.5 × 10-9. Calculate the solubility of barium sulphate in pure water and in 0.1 M BaCl2. [2]
(c) Explain the following:
(i) When NH4Cl and NH4OH are added to a solution containing both, Fe3+ and Ca2+ ions, which ion is precipitated first and why? [2]
(ii) Dissociation of H2S is suppressed in acidic medium. [1]
Answer:
(c) (i) Addition of NH4Cl decreases the dissociation of weak base NH4OH due to the common ion effect.
The concentration of OH– is small but the ionic product [Fe3+] [OH3-] exceeds the K of metal hydroxide of group III and they get precipitated.
(ii) Only Cu2+ and Pb2+ are precipitated as CuS and PbS respectively. These are the metal sulphides of group II. The Ks of these metal sulphides is small. Due to common ion (H+) effect, the dissociation of wealth acid H2S is suppressed.
Section – B
(Answer any two questions)
Question 5.
(a) Write the IUPAC names of the following coordination compounds: [1]
(i) [Cr(NH3)4(H2O)2]Cl3
(ii) [PtCl2(NH3)4] [PtCl4]
(b) State the hybridization and magnetic property of [Fe(CN)6]3- ion according to the valence bond theory. [1]
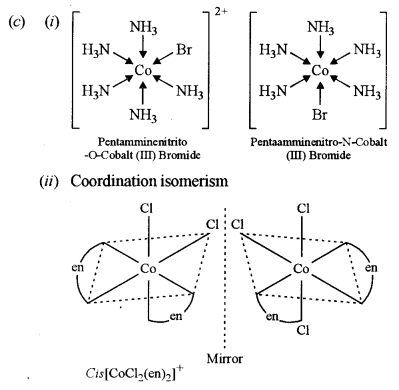
(c) (i) What type of isomers are [Co[NH3)3Br]SO4 and [Co(NH3)5SO4]Br? Give a chemical test to distinguish between them.
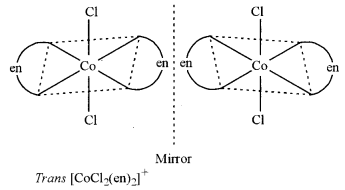
(ii) Write the structures of optical isomers of the complex ion [Co(en)2Cl2]+ [1]
Answer.
(a) (i) Tetraamminediquachromium (III) chloride
(ii) Tetraamminedichloridoplatinum (IV) tetra chloro platinate (II)
(b) Fe3+ has 6 vacant d2sp3 hybrid orbitals. The hybrid orbitals accommodate 6 pairs of electrons from 6CN– ions and form 6 coordinate bonds. Now, Fe3- has one unpaired electron and, therefore, [Fe(CN)6]4- ion is paramagnetic.
Question 6.
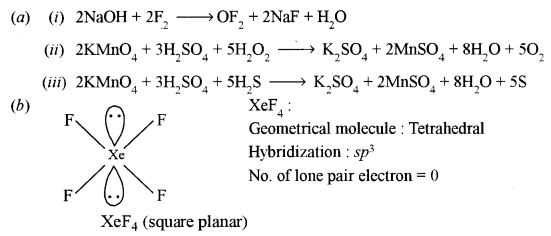
(a) Give balanced chemical equations for the following reactions: [3]
(i) Fluorine is passed through a cold, dilute NaOH solution.
(ii) Hydrogen peroxide is treated with acidified KMnO4 solution.
(iii) Sulphuric acid is treated with hydrogen sulphide.
(b) Draw the structure of xenon tetrafluoride molecule and state the hybridization of the central atom and the geometry of the molecule. [2]
Answer:
Question 7.
(a) Name the important ore of silver. Write all the steps and reactions involved in the cyanide process for the extraction of silver from its ore. [3]
(b) Explain the following: [2]
(i) Why do transition metal ions possess a great tendency to form complexes?
(ii) The paramagnetic character in 3d-transition series elements increases up to Mn and then decreases.
Answer:
(a) Important ore of silver is silver glance -Ag2S, Pyragyrite – 3AgS.Sb2S3, Horn silver – AgCl
Silver nitrate reaction with KCN solution white ppt. of silver cyanide is fonned which dissolves in excess of KCN due to complex formation.
\(\begin{aligned} \mathrm{AgNO}_{3}+\mathrm{KCN} & \longrightarrow \mathrm{AgCN}+\mathrm{KNO}_{3} \\ \mathrm{AgCN}+\mathrm{KCN} & \longrightarrow \mathrm{K}\left[\mathrm{Ag}(\mathrm{CN})_{6}\right] \end{aligned}\)
Extraction of silver by cyanide process:
(i) Concentration: The sulphide ore is crushed, powdered and then concentrated by the froth floatation process.
(ii) Treatment with Sodium Cyanide:
\(\begin{array}{l}{\left.\mathrm{Ag}_{2} \mathrm{S}+4 \mathrm{NaCN} \rightleftharpoons 2 \mathrm{Na}[\mathrm{AgCN}]_{2}\right]+\mathrm{Na}_{2} \mathrm{S}} \\ {\mathrm{Na}_{2} \mathrm{S}+2 \mathrm{O}_{2}+\mathrm{H}_{2} \mathrm{O} \longrightarrow \mathrm{Na}_{2} \mathrm{S}_{2} \mathrm{O}_{3}+2 \mathrm{NaOH}} \\ {\mathrm{Na}_{2} \mathrm{S}_{2} \mathrm{O}_{3}+2 \mathrm{NaOH}+\mathrm{O}_{2} \longrightarrow \mathrm{Na}_{2} \mathrm{SO}_{4}+\mathrm{H}_{2} \mathrm{O}}\end{array}\)
(iii) Precipitation of silver:
\(2 \mathrm{Na}\left[\mathrm{Ag}(\mathrm{CN})_{2}\right]+\mathrm{Zn} \longrightarrow \mathrm{Na}_{2}\left[\mathrm{Zn}(\mathrm{CN})_{4}\right]+2 \mathrm{Ag} \downarrow\)
(iv) Fusion: The precipitate silver is filtered, pressed, dried and fused with borax when a bright compact mass is obtained.
(v) Purification: Silver usually contains impurities of Zn, Cu and Au. This purified by electrolysis process. Thus, we get pure silver.
(b) (i) Because cations of transition elements have vacant d-orbitals of appropriate energy and these cations have high +ve charge and small size.
(ii) Because magnetic moment increased upto Mn and then decreases.
Section – C
(Answer any two questions)
Question 8.
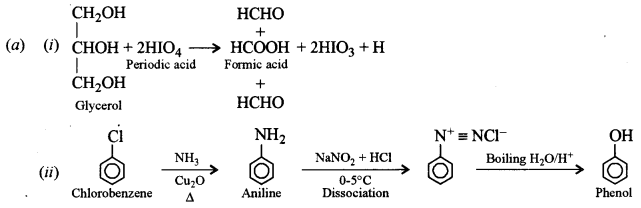
(a) How can the following conversions be brought about:
(i) Glycerol to formic acid [1]
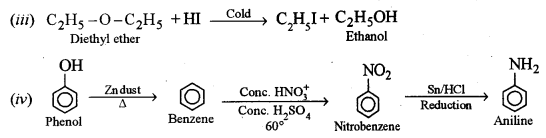
(ii) Chlorobenzene to phenol [1]
(iii) Diethyl ether to ethanol [1]
(iv) Phenol to aniline. [2]
(b) (i) How is iodoform prepared from ethanol? Give a balanced equation. [1]
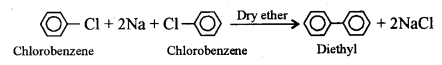
(ii) What will be the product formed when chlorobenzene is heated with sodium metal in the presence of dry ether? [1]
(c) Identify the compounds A, B, C, D, E and F: [3]
![]()
Answer:
(b) (i) Ethyl alcohol when heated with iodine and sodium hydroxide or aqueous sodium carbonate forms a yellow crystalline solid, iodoform.
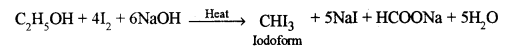
(ii) When chlorobenzene is heated with sodium metal in presence of dry ether, the chlorine atom of chlorobenzene is replaced by the phenol group and diphenyl is obtained. This is called Fitting reaction.
(c) A – Acetic acid and propanoic acid
B – 2, 2 – Dichloropropane
C – CH3COONH4
D – CH3CH2OH
E – CH3OH
F – HCHO
Question 9.
(a) Give balanced equations for the following name reactions: [3]
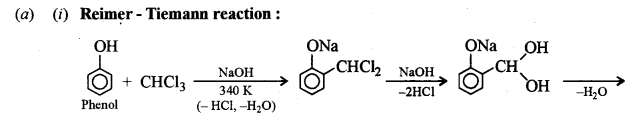
(i) Reimer-Tiemann reaction.
(ii) Rosenmund reaction
(iii) Hoffmann s degradation reaction
(b) Give one chemical test to distinguish between the following pairs of compounds: [3]
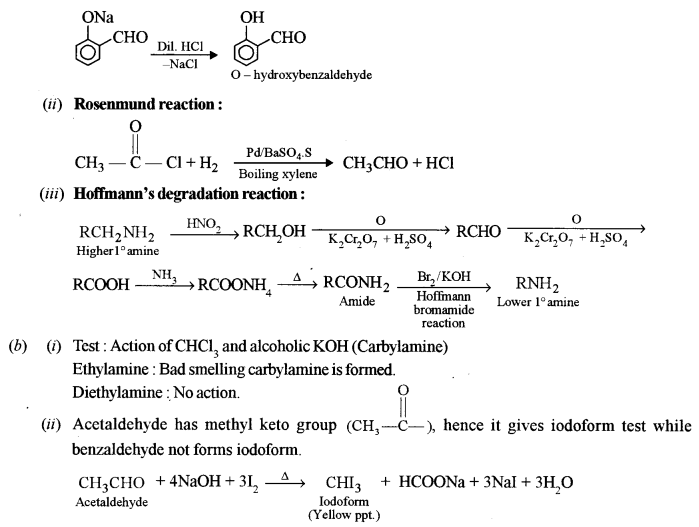
(i) Ethylamine and diethylamine
(ii) Acetaldehyde and benzaldehyde
(c) (i) Arrange the following compounds in the ascending order of their basic strength and give reasons for your answer: [2]
Methylamine, Aniline, Ethylamine, Diethyl ether
(ii) Name the monomers and the type of polymerization in each of the following polymers: [2]
(i) Polyester
(ii) Bakelite
Answer:
(c) (i) Aniline, Diethyl ether, methylamine, ethylamine
(ii) (a) Polyester – Synthetic polymers
(b) Bakelite, Thermoplastics polymer
Question 10.
(a) An organic compound A with molecular formula C2H7N on reaction with nitrous acid gives a compound B. B on controlled oxidation gives compound C. C reduces Tollens reagent to give silver mirror and D. B reacts with D in the presence of concentrated sulphuric acid to give sweet-smelling compound E. Identify A, B, C, D and E. Give the reaction of C with ammonia. [3]
(b) Give balanced equations for the following reactions: [4]
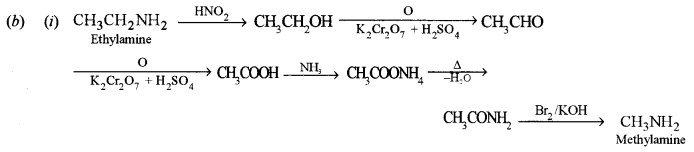
(i) How will you convert ethyl amine to methylamine?
(ii) What is the effect of denaturation on the structure of proteins?
(iii) Name the nitrogen base residues present in DNA.
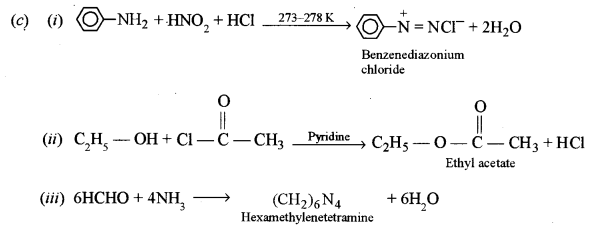
(c) Give balanced equations for the following reactions: [3]
(i) Aniline is treated with nitrous acid and HCl at low temperature.
(ii) Acetyl chloride is treated with ethyl alcohol.
(iii) Formaldehyde is treated with ammonia.
Answer:
(a) A – Ethylamine (ethanamine)
B – CH3CH2OH (ethyl alcohol)
C – CH3CHO (acetaldehyde)
D – CH3COOH (acetic acid)
E – CH3COOC2H5 (ethyl acetate)
(ii) When the soluble form of proteins such as globular proteins is subjected to the action of heat, acids, alkalies or electrolytes, they undergo coagulation to form fibrous proteins which are insoluble in water. This results in to change of physical and biological properties of the proteins. The coagulated protein thus obtained is called denatured protein.
(iii) Nitrogen base residue present in DNA:
Purine bases: Adenine, Guanine
Pyrimidine bases: Cytosine, Thymine.