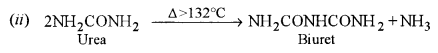ISC Chemistry Previous Year Question Paper 2014 Solved for Class 12
Maximum Marks: 70
Time allowed: 3 hours
- Answer all questions in Part I and six questions from Part II, choosing two questions from Section A, two from Section B and two from Section C.
- All working, including rough work, should be done on the same sheet as, and adjacent to, the rest of the answer.
- The intended marks for questions or parts of questions are given in brackets [ ].
- Balanced equations must be given wherever possible and diagrams where they are helpful.
- When solving numerical problems, all essential working must be shown.
- In working out problems use the following data:
Gas constant R = 1.987 cal deg-1 mol-1 = 8.314 JK-1 mol-1 = 0.0821 dm3 atm K-1 mol-1. 1L atm = 1 dm3 atm = 101.3 J.
1 Faraday = 96500 Coulombs, Avogadro’s number = 6.023 × 1023
Part – I
(Answer all questions)
Question 1.
(a) Fill in the blanks by choosing the appropriate word/words from those given in the brackets: [5]
(increases, formic acid, decreases, less, zero, small, paired, atoms, unpaired, ions, pentagonal bipyramidal, electrical, more, ethylamine, molecules, propanoic acid, methylamine, chemical.)
(i) An electrochemical cell converts ……… energy to ……….. energy.
(ii) The crystal of graphite is made-up of ………. while that of sodium chloride is made-up of ………..
(iii) Ethyl isocyanide, on hydrolysis with dilute sulphuric acid, gives ……… and ………
(iv) The molar conductance of a solution ………… with dilution, while its specific conductance ……….. with dilution.
(v) The Van’t Hoff factor of the acetic acid solution is …………… than one and the value of a normal colligative property is ……… than the observed colligative property of this solution.
(b) Complete the following statements by selecting the correct alternative from the choices given: [5]
(i) Of the following terms used for denoting the concentration of a solution, the one which does not get affected by temperature is:
(1) Molarity
(2) Molality
(3) Normality
(4) Formality
(ii) The solubility of calcium hydroxide is s mol litre-1. The solubility product under the same condition will be:
(1) 4s3
(2) 2s3
(3) 2s2
(4) s3
(iii) A current liberates 0.50 g of hydrogen in 2 hours. The weight of copper (at. wt. = 63.5) deposited at the same time by the same current through copper sulphate solution is:
(1) 63.5 g
(2) 31.8 g
(3) 15.9 g
(4) 15.5 g
(iv) Natural rubber is a:
(1) Polyester
(2) Polyamide
(3) Polyisoprene
(4) Polysaccharide
(v) Among the following halogens, the one which does not form an oxyacid is:
(1) Fluorine
(2) Chlorine
(3) Bromine
(4) Iodine
(c) Answer the following questions: [5]
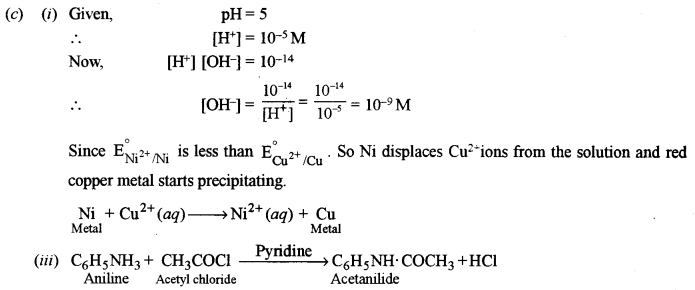
(i) What is the [OH–] concentration of an acid whose pH is 5 at 25°C?
(ii) What happens when a nickel rod is dipped into a copper sulphate solution? Justify your answer.
\(\left[\mathrm{E}_{\mathrm{Ni}^{2+} / \mathrm{Ni}}^{\circ}=-0.25 \mathrm{V} \text { and } \mathrm{E}_{\mathrm{Cu}^{2+} / \mathrm{Cu}}^{\circ}=0.34 \mathrm{V}\right]\)
(iii) Write the equation for the preparation of acetanilide from aniline.
(iv) Define Raoult’s law for the elevation of the boiling point of a solution.
(v) An ionic compound is made-up of A cations and B anions. If A cations are present at the alternate comers and B anion is present on the body of the diagonal, what is the formula of the ionic compound? [5]
(d) Match the following:
| (i) Molal depression constant | (a) Infinite dilution |
| (ii) Acetaldehyde | (b) mol L-1 sec-1 |
| (iii) Rate of reaction | (c) Iodoform |
| (iv) Optical activity | (d) K kg mol-1 |
| (v) Kohlrausch’s law | (e) Lactic acid |
Answer:
(a) (i) chemical, electrical
(ii) atoms, ions
(iii) ethylamine, formic acid
(iv) increases, decreases
(v) more, less
(b) (i) (2) Molality
(ii) (1) 453
(iii) (3) 15.9 g
(iv) (3) Polyisoprene
(v) (1) Fluorine
(iv) According to Raoult’s law: ‘Elevation of a boiling point of a solution is directly proportional to the lowering in vapour pressure caused by the number of particles of solute present in the solution’.
(v) The formula of an ionic compound is A+B– or AB.
(d) (i) (d)
(ii) (c)
(iii) (b)
(iv) (e)
Part – II
Answer six questions choosing two from Section A, two from Section B and two from Section C.
Section – A
(Answer any two questions)
Question 2.
(a) (i) What will be the vapour pressure of a solution containing 5 moles of sucrose (C12H22O11) in 1 kg of water, if the vapour pressure of pure water is 4.57 mm of Hg? [C = 12, H = 1, O = 16] [3]
(ii) A 2 molal solution of sodium chloride in water causes an elevation in the boiling point of water by 1.88 K. What is the value of Vant Hoff factor? What does it signify? [Kb = 0.52 K kg mol-1] [2]
(b) (i) Write the mathematical expression relating the variation of the rate constant of a reaction with temperature. [4]
(ii) How can you graphically find the activation energy of the reaction from the above expression?
(iii) The slope of the line in the graph of log k (k = rate constant) versus \(\frac { 1 }{ T }\) is -5841. Calculate the activation energy of the reaction.
(c) Define the Frenkel defect in a solid crystal. [1]
Answer:
(a) (i) According to Raoult’s law, for a solution containing non-volatile solute

This value of ‘i’ signifies that when 1 mole of NaCl is dissolved in water it dissociates to give 1.8076 moles of particles in the solution.
(b) (i) The mathematical expression of the Arrhenius equation:
\(k=\mathrm{A} e^{-\mathrm{E}_{a} / \mathrm{RT}}\)
where k is rate constant of the given reaction
A is another constant called frequency factor
Ea is activation energy
R is gas constant
T is the temperature in Kelvin.
(ii) Finding activation energy graphically: Taking a log of Arrhenius equation
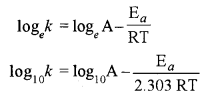
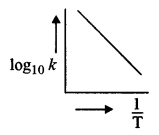
It is an equation for a straight line (y = mx + C)
Therefore, on plotting a graph between different values of log10k and \(\frac { 1 }{ T }\) at different temperature, a straight line as shown in fig. is obtained and slope of line is equal to \(-\frac{\mathrm{E}_{a}}{2.303 \mathrm{R}}\)
Thus, by knowing slope, we can calculated Ea
(iii) \(\text { Slope }=-\frac{\mathrm{E}_{a}}{2.303 \mathrm{R}}\)
or -Ea = -5841 × 2.303 × 8.314
Ea = 111838.45 J mol-1 = 111.838 kJ mol-1
(c) Frenkel defect: When some ions (usually cations) are missing from the lattice sites and occupy interstitial sites so that electrical neutrality as well as stoichiometry is maintained, it is called Frenkel defect.
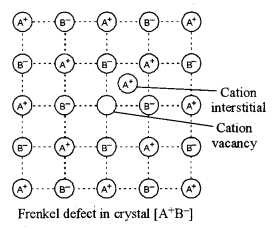
Question 3.
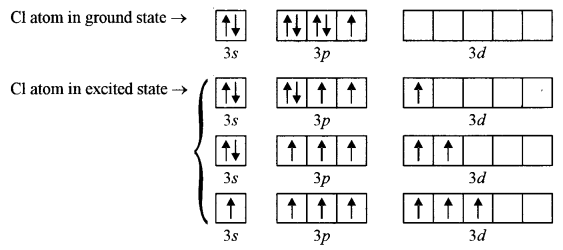
(a) Explain giving reasons why: [4]
(i) Ionic solids conduct electricity in the molten state, but not in solid-state.
(ii) A solution of sodium chloride has no effect on litmus, but a solution of zinc chloride turns blue litmus red.
(b) In a crystal of diamond: [2]
(i) How many carbon atoms are present per unit cell?
(ii) What type of lattice does diamond crystallize in?
(iii) How many carbon atoms surround each carbon atom?
(iv) How are they arranged?
(c) (i) What is a standard hydrogen electrode? [1]
(ii) 0.05 M NaOH solution offered resistance of 31.6 ohms in a conductivity cell at 298 K. If the cell constant of the cell is 0.367 cm-1, calculate the molar conductivity of the NaOH solution. [3]
Answer:
(a) (i) In solid-state of a substance (ionic compound) ions are not free to move but in the molten (liquid) state they are free to move towards oppositely charged electrodes on passing an electric current. Hence, an ionic substance conducts electricity in the molten state and not in the solid-state.
(ii) Sodium chloride does not undergo hydrolysis when dissolved in water, as it is salt of strong acid and a strong base and solution remain neutral. Zinc chloride is a salt of strong acid and weak base so it undergoes hydrolysis in water (cationic hydrolysis) forming less dissociated Zn(OH)2 (a weak base) and strong acid HCl which is almost completely dissociated. Therefore, the concentration of H+ ions increases and the solution becomes acidic and turns blue litmus red.
\(\begin{array}{c}{\mathrm{NaCl}+\mathrm{H}_{2} \mathrm{O} \rightleftharpoons \mathrm{Na}^{+}+\mathrm{Cl}^{-}+\mathrm{H}^{+}+\mathrm{OH}^{-}} \\ {\mathrm{ZnCl}_{2}+2 \mathrm{H}_{2} \mathrm{O} \rightleftharpoons \mathrm{Zn}(\mathrm{OH})_{2}+2 \mathrm{H}^{+}+2 \mathrm{Cl}^{-}}\end{array}\)
(b) (i) Four carbon atoms are present per unit cell.
(ii) Diamond crystallizes in a network type of lattice with free arrangement of carbon atoms.
(iii) Four carbon atoms surround each carbon atom.
(iv) Four carbon atoms are tetrahedrally arranged around each central carbon atom.
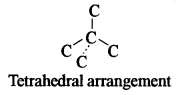
(c) (i) Standard Hydrogen electrode: It is a reference electrode used to measure the electrode potentials of the electrodes.
In this hydrogen gas at 1 atm. pressure is passed into HCl at 25°C in which a Pt foil coated with finely divided platinum black remains immersed. The electrode potential of SHE is taken as zero at 298 K.
(ii) Given, Molarity of NaOH solution = 0.05 M
Resistance = 31.6 ohm
Cell constant = 0.367 cm-1

Question 4.
(a) (i) Kc for the reaction \(\mathrm{SO}_{2(g)}+\frac{1}{2} \mathrm{O}_{2(g)} \rightleftharpoons\) is 61.7 at 60°C. What is its unit? Calculate Kp for the reaction and write its unit. [3]
(ii) What happens to the equilibrium in a reversible reaction if a catalyst is added to it? [1]
(b) State the effect of the following on the reaction \(2 \mathrm{SO}_{2(\mathrm{g})}+\mathrm{O}_{2(\mathrm{g})} \rightleftharpoons 2 \mathrm{SO}_{3(\mathrm{g})}+189.4 \mathrm{kJ}\) at equilibrium: [2]
(i) Temperature is increased.
(ii) The concentration of SO2 is increased.
(iii) The pressure is decreased.
(iv) Helium is added at constant pressure.
(c) (i) 0.3605 g of a metal is deposited on the electrode bypassing 1.2 amperes of current for 15 minutes through its salt solution. The atomic weight of the metal is 96. What is the valency of the metal? [3]
(ii) Explain why phenolphthalein is used as an indicator in acid-base titration. [1]
Answer:
(a) (i) For given reaction unit of Kc is L mol-1 and with given coefficients, the unit is (L mol-1)1/2
Kp = Kc (RT)Δn
For given reaction, Kc = 61.7, R = 0.0821, T = 60 + 273 = 333 K
and Δn = number of moles of gaseous product – Number of moles of gaseous reactants
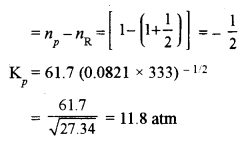
Unit of Kp is atm and for given coefficients unit of Kp is atm1/2.
(ii) On addition of a catalyst to a reversible reaction, the equilibrium remains unaffected but is attained quickly.
(b) (i) As a balanced reaction is endothermic, equilibrium will shift in a backward direction with an increase in temperature.
(ii) Increase in conc, of reactants, shift the equilibrium in the forward direction, therefore, upon increasing conc. of SO2 equilibrium will shift in the forward direction or more of SO3 will be formed.
(iii) As backward reaction proceeds with the increase in the number of moles, the backward reaction will be favoured.
(iv) The backward reaction will be favoured.
(c) (i) Weight ‘w’ of the metal = 0.3605 g
Current = 1.2 amp.
Time = 15 min = 15 × 60 sec.
The atomic weight of metal = 96
According to Faraday’s first law of electrolysis,

Valency of metal is 3.
(ii) Phenolphthalein is a complex weak organic acid (HPh). Its undissociated molecule (HPh) is colourless while its ion (Ph-) is pink in colour. pH range of phenolphthalein is 8 – 9.8.
In acid-base titration as the medium changes from acidic to basic, the following equilibrium shifts to right.
\(\mathrm{HPh} \rightleftharpoons \mathrm{H}^{+}+\mathrm{Ph}^{-}\) and solution turns pink from colourless giving its endpoint. When medium changes to acidic, equilibrium shifts to left and the solution becomes colourless.
Section – B
(Answer any two questions)
Question 5.
(a) Write the formula of the following compounds: [2]
(i) Triamminetriaquachromium (III) chloride.
(ii) Potassium hexacyanoferrate (III).
(b) Name the types of isomerism shown by the following pairs of compounds: [2]
(i) [CoCl (H2O)](NH3)4Cl2 and [CoCl2(NH3)4] ClH2O
(ii) [Pt(NH3)4] [PtCl6] and [Pt(NH3)4Cl2] [PtCl4]
(c) For the complexion of [Co(NH3)6]3+: [1]
(i) State the hybridisation of the complex.
(ii) State the magnetic nature of the complex.
Answer:
(a) (i) [Cr(NH3)3(H2O)3]Cl3
(ii) K3[Fe(CN)6]
(b) (i) Hydrate isomerism
(ii) Coordination isomerism.
(c) (i) Complex has d2sp3 hybridisation.
(ii) Complex is diamagnetic in nature.
Question 6.
(a) Write balanced chemical equations for the following reactions: [3]
(i) Ozone and lead sulphide.
(ii) Chlorine is passed through hot concentrated NaOH solution.
(iii) Sulphuric acid is treated with phosphorus.
(b) Give reasons for the following: [2]
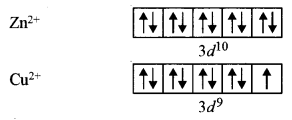
(i) Zn2+ salts are white but Cu2+ salts are blue in colour.
(ii) Fluorine gives only one oxide but chlorine gives a series of oxides.
Answer:
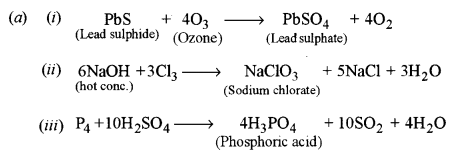
(b) (i) Transition metals and their ions are generally coloured due to d-d transition of the unpaired electron.
But is the case of Zn2+ 3d is completely filled and there is no unpaired electron available for transition. So light falling gets completely transmitted and, therefore, Zn2+ salts appear white. However, in Cu2+ 3d has 9 electrons. The unpaired electron absorbs particular wavelengths in the visible region of light and the transmitted light shows the complementary colour ‘blue’.
(ii) The valence shell electronic configuration of fluorine is 2s22p5 and there are no vacant d orbitals. So it cannot form more than one oxide i.e. OF2.
Chlorine, on the other hand, has 3 vacant 3d orbitals in which electrons can be excited. Therefore, it can show variable covalency and give a series of oxides. Like Cl2O, ClO2 etc.
Question 7.
(a) How is potassium dichromate prepared from a sample of chromite ore? Give balanced equations for the chemical reactions involved. [3]
(b) For the molecule IF7:
(i) Draw the structure of the molecule.
(ii) State the hybridisation of the central atom.
(iii) State the geometry of the molecule. [2]
Answer:
(a) Preparation of potassium dichromate from chromite ore involves the following steps:
(i) Conversion of chromite ore to sodium chromate: It is achieved by roasting finely powdered chromite ore with sodium carbonate and quick lime in excess of air.
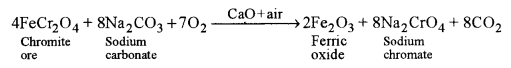
(ii) Conversion of sodium chromate to sodium dichromate: It is achieved by the action of conc. H2SO4 on sodium chromate solution.
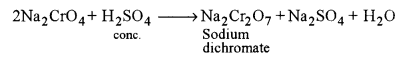
(iii) Conversion of sodium dichromate to potassium dichromate: It is achieved by treating hot conc. solution of sodium dichromate with KCl.
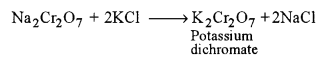
The resulting solution on cooling yields orange crystals of potassium dichromate.
(b) (i)
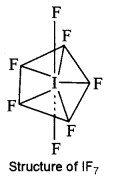
(ii) The hybridisation of I is sp3d3
(iii) Geometry is pentagonal bipyramidal.
Section – C
(Answer any two questions)
Question 8.
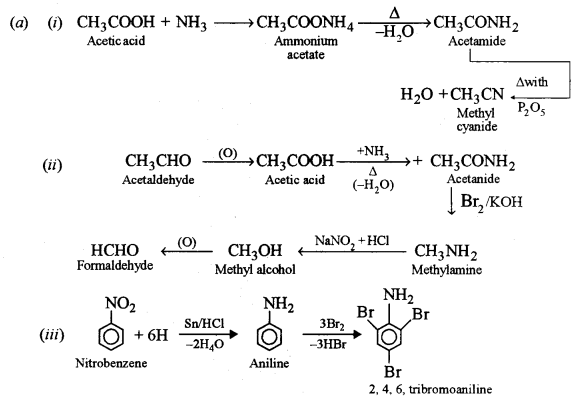
(a) How can the following conversions be brought about:
(i) Acetic acid to methyl cyanide.
(ii) Acetaldehyde to formaldehyde.
(iii) Nitrobenzene to 2, 4, 6 – tribromoaniline.
(b) Identify the reagents A, B, C, D, E and F required for the following conversion: [3]
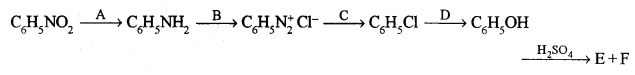
Answer:
(b) A → Sn + conc. HCl
B → NaNO2 + conc. HCl
C → Cu2Cl2 + HCl
D → NaOH
E → 2-hydroxy benzene sulphonic acid.
F → 4-hydroxy benzene sulphonic acid.
Question 9.
(a) The deficiency of which vitamin will cause the following diseases:
(i) Scurvy
(ii) Haemorrhages
(b) Give one chemical test to distinguish between the following pairs of compounds:
(i) Ethanol and 2 propanols.
(ii) Aniline and ethylamine.
(c) Write the structures of all enantiomers possible for lactic acid.
(d) Give balanced equations for the following reactions:
(i) Acetaldehyde is heated with hydroiodic acid in the presence of red phosphorus.
(ii) Calcium acetate is subjected to dry distillation.
(iii) Sodium ethoxide is treated with ethyl bromide.
(iv) Benzaldehyde is treated with sodium bisulphite.
Answer:
(a) (i) Vitamin C
(ii) Vitamin K
(b) (i)
| Ethanol | 2-Propanol |
| When treated with Lucas reagent (anhy. ZnCl2 and cone. HCl), ethanol gives turbidity after heating. | When treated with Lucas reagent, 2-propanol gives turbidity within five minutes without heating. |
(ii) Aniline and ethylamine can be distinguished using the azo-dye test.
| Aniline | Ethylamine |
| Aniline is treated with HN02 (NaNO2 + dil. HCl) at 0-5°C to give’ benzene diazonium chloride, which on further treatment with an alkaline solution of (3-naphthol gives red dye. | Ethylamine gives a brisk effervescence due to evolution of N2 gas under similar conditions. |
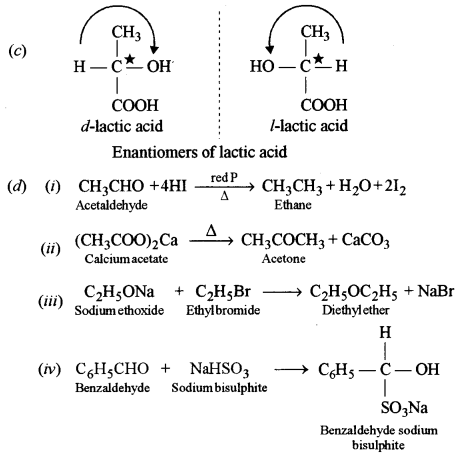
Question 10.
(a) An organic compound A with the molecular formula C7H8 on oxidation by chromyl chloride in the presence of CCl4 gives a compound B which gives positive tollen’s test. The compound B on treatment with NaOH followed by acid hydrolysis gives two products C and D. C on oxidation gives B which on further oxidation gives D. The compound D on distillation with soda lime gives a hydrocarbon E. Below 60°C, concentrated nitric acid reacts with E in the presence of concentrated sulphuric acid forming a compound F.
Identify the compounds A, B, C, D, E and F. [3]
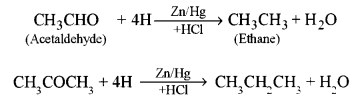
(b) Give balanced equations for the following name reactions: [3]
(i) Clcmmensen’s reduction.
(ii) Kolbe’s electrolytic reaction.
(iii) Balz-Schiemann’s reaction.
(c) (i) What do you observe when glucose is treated with bromine water? [2]
(ii) What is the isoelectric point?
(d) Answer the following: [2]
(i) What is the biuret test?
(ii) Write a balanced equation for the formation of biuret.
Answer:
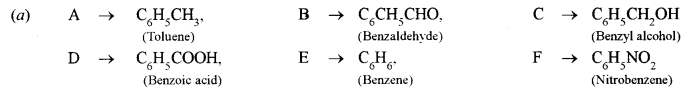
(b) (i) Clemmensen’s reduction: It is the reduction of aldehydes and ketones to alkanes with amalgamated zinc and conc. HCl.
(ii) Kolbe’s electrolytic reaction: In this reaction, the aqueous solution of the sodium salt of the carboxylic acid is electrolyzed to obtain alkane, alkene or alkyne.
e.g., Ethane is framed sodium acetate.
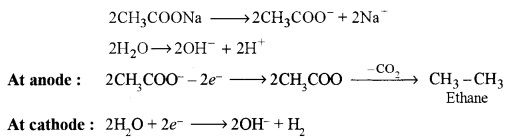
(iii) Balz – Schiemann reaction: In this reaction, aniline is first converted to benzene diazonium fluoroborate which on heating gives fluorobenzene.
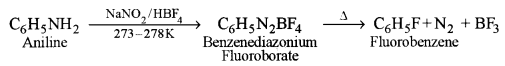
(c) (i) When glucose is treated with bromine water, bromine water gets decolourised.
(ii) Isoelectric point: It is the pH at which there is no net migration of an amino acid under the influence of an applied electric field.
(d) (i) Biuret test: When urea is heated just above its melting point it forms a compound called biuret which gives a violet colour with dilute copper sulphate solution in alkaline medium.