ISC Chemistry Previous Year Question Paper 2013 Solved for Class 12
Maximum Marks: 70
Time allowed: 3 hours
- Answer all questions in Part I and six questions from Part II, choosing two questions from Section A, two from Section B and two from Section C.
- All working, including rough work, should be done on the same sheet as, and adjacent to, the rest of the answer.
- The intended marks for questions or parts of questions are given in brackets [ ].
- Balanced equations must be given wherever possible and diagrams where they are helpful.
- When solving numerical problems, all essential working must be shown.
- In working out problems use the following data:
Gas constant R = 1.987 cal deg-1 mol-1 = 8.314 JK-1 mol-1 = 0.0821 dm3 atm K-1 mol-1. 1L atm = 1 dm3 atm = 101.3 J.
1 Faraday = 96500 Coulombs, Avogadro’s number = 6.023 × 1023
Part – I
(Answer all questions)
Question 1.
(a) Fill in the blanks by choosing the appropriate word/words from those given in the brackets: [5]
(zero, first, second, increased, decreased, anode, cathode, active, inactive, potassium cyanide, internal, external, dependent, independent, red, benzoic acid, benzoin, common ion effect, salt hydrolysis, alkali, potassium hydroxide.)
(i) In a galvanic cell, electrons flow from ………. to ……….. through the connecting wires.
(ii) Racemic mixtures are optically …………. because of …………… compensation.
(iii) The half-life period of a …………. order reaction is …………. of the concentration of the reactant.
(iv) Benzaldehyde when treated with an alcoholic solution of ……… forms ………..
(v) The solubility of calcium oxalate is …………. in the presence of ammonium oxalate because of ……….
(b) Complete the following statements by selecting the correct alternative from the choices given: [5]
(i) The compound which is optically active is:
(1) 1-butanol
(2) 2-butanol
(3) 1-propanol
(4) 2-methyl-l-propanol
(ii) The salt which will not hydrolyse in aqueous solution is:
(1) Copper sulphate
(2) Sodium sulphate
(3) Potassium cyanide
(4) Sodium carbonate
(iii) Copper has the face centred cubic structure. The coordination number of each ion is:
(1) 4
(2) 12
(3) 14
(4) 8
(iv) For the reaction \(2 \mathrm{SO}_{2}+\mathrm{O}_{2} \rightleftharpoons 2 \mathrm{SO}_{3}\) the unit of equilibrium constant is:
(1) L mol-1
(2) J mol-1
(3) mol L-1
(4) [L mol-1]2
(v) The deficiency of vitamin D causes:
(1) Rickets
(2) Gout
(3) Scurvy
(4) Night blindness
(c) Answer the following questions: [5]
(i) Two metallic elements A and B have the following standard oxidation potentials:
A = 0.40 V, B = -0.80 V. What would you expect if element A was added to an aqueous salt solution of element B? Give a reason for your answer.
(ii) Two moles of NH3 are introduced into the one-litre flask in which it dissociates at high temperature as follows:
\(2 \mathrm{NH}_{3}(g) \rightleftharpoons \mathrm{N}_{2}(g)+3 \mathrm{H}_{2}(g)\)
Determine Kc if at equilibrium 1 mole of NH3 remains.
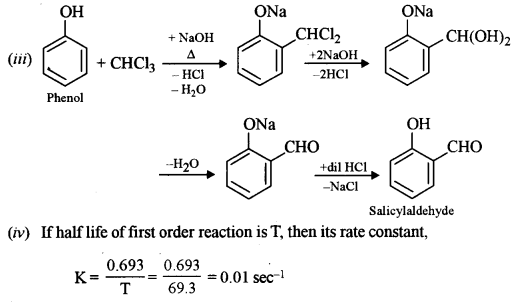
(iii) Give a balanced equation for the preparation of salicylaldehyde from phenol.
(iv) If the half-life period for a first-order reaction is 69.3 seconds, what is the value of its rate constant?
(v) Define cryoscopic constant.
(d) Match the following: [5]
| (i) Colligative property | (a) Polysaccharide |
| (ii) Nicol prism | (b) Osmotic pressure |
| (iii) Activation energy | (c) Aldol condensation |
| (iv) Starch | (d) Polarimeter |
| (v) Acetaldehyde | (e) Arrhenius equation |
Answer:
(a) (i) anode, cathode
(ii) inactive, external
(iii) first, independent
(iv) potassium cyanide, benzoin
(v) decreased, common ion effect
(b) (i) (2) 2-butanol
(ii) (2) Sodium sulphate
(iii) (2) 12
(iv) (1) L mol-1
(v) (1) Rickets
(c) (i) Since the oxidation potential of A is higher than that of B, therefore, metal A will displace metal B from its salt solution.
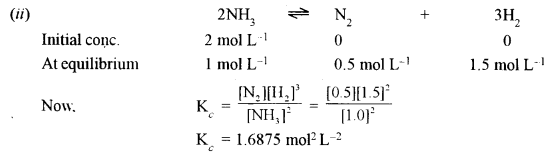
(v) Cryoscopic constant: It is defined as the depression in freezing point of the solvent produced on dissolving one mole of a substance in 1000 g of it. It is also called molal depression constant.
(d) (i) Colligative property → (b) Osmotic pressure
(ii) Nicol prism → (d) Polarimeter
(iii) Activation energy → (e) Arrhenius equation
(iv) Starch → (a) Polysaccharide
(v) Acetaldehyde → (c) Aldol condensation
Part – II
Answer six questions choosing two from Section A, two from Section B and two from Section C.
Section – A
(Answer any two questions)
Question 2.
(a) (i) Ethylene glycol is used as an antifreeze agent. Calculate the amount of ethylene glycol to be added to 4 kg of water to prevent it from freezing at -6°C. (Kf for H2O = 1.85 K mole-1 kg) [3]
(ii) The freezing point of a solution containing 0.3 g of acetic acid in 30 g of benzene is lowered by 0.45 K. Calculate the Van’t Hoff factor.
(at. wt. of C = 12, H = 1, O = 16, Kf for benzene = 5.12 K kg mole-1) [2]
(b) Name the law or principle confirmed by the following observations:
(i) When water is added to 0.01 M aqueous solution of acetic acid the number of hydrogen ions increase. [2]
(ii) When 96500 coulombs of electricity is passed through acidulated water, 5.6 litres of oxygen at s.t.p. is liberated at the anode. [1]
(c) Arrange Ag, Cr and Hg metals in the increasing order of reducing power. Given: [1]
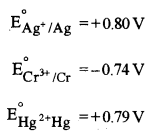
(d) In a first-order reaction, 10% of the reactant is consumed in 25 minutes. Calculate: [2]
(i) The half-life of the reaction.
(ii) The time required for completing 17% of the reaction.
Answer:



Question 3.
(a) Explain giving reasons why (Give equations in support of your answer):
(i) A solution of NH4Cl and NH4OH acts as a buffer. [2]
(ii) Cu is precipitated as CuS while Zn is not precipitated when H2S is passed through an acidic solution of Cu(NO3)2 and Zn(NO3)2 respectively. [2]
(b) (i) What is Schottky defect in a solid? [1]
(ii) A bcc element (atomic mass 65) has a cell edge of 420 pm. Calculate its density in g/cm3 [3]
(c) The rate of the reaction \(\mathrm{H}_{2}+\mathrm{I}_{2} \rightleftharpoons 2 \mathrm{HI}\) is given by:
Rate = 1.7 × 10-19 [H2] [I2] at 25°C. [2]
The rate of decomposition of gaseous HI to H2 and I2 is given by:
Rate = 2.4 × 10-21 [HI]2 at 25°C.
Calculate the equilibrium constant for the formation of HI from H2 and I2 at 25°C. [2]
Answer:
(a) (i) NH4Cl is a strong electrolyte hence, dissociates completely. NH4OH is a weak electrolyte and dissociates to a small extent. Its dissociation is further suppressed by common ion \(\mathrm{NH}_{4}^{+}\) provided by NH4Cl in the solution.
\(\begin{array}{c}{\mathrm{NH}_{4} \mathrm{Cl}_{(a q)} \rightleftharpoons \mathrm{NH}_{4(a q)}^{+}+\mathrm{Cl}_{\max }^{-}} \\ {\mathrm{NH}_{4} \mathrm{OH}_{(a q)} \Longrightarrow \mathrm{NH}_{4(a q)}^{+}+\mathrm{OH}_{(a q)}^{-}}\end{array}\)
This solution acts as a basic buffer and maintains its pH around 9.25. It resists the change in pH on the addition of a small amount of acid or alkali. This can be explained as below:
Upon adding a small amount of HCl to this solution, H+ ions of HCl gets neutralised by OH– ions already present and more of NH4OH molecules get ionised to compensate for the loss of OH– ions. Thus, pH practically remains unchanged.
Upon adding a small amount of NaOH to this solution, OH ions of NaOH combine with \(\mathrm{NH}_{4}^{+}\) ions already present to form weakly ionised NH4OH. Thus, pH of the solution remains practically unchanged.
(ii) This is because the Ksp of CuS is very low as compared to that of ZnS. Therefore, a low concentration of S2- ions can precipitate Cu2+ ions as CuS, whereas a high concentration of S2 ions is required to precipitate Zn2+ ions as ZnS.
Now, in the presence of HCl, dissociation of H2S is suppressed due to the common ion effect.
HCl → H+ + Cl– (strong electrolyte)
H2S → 2H+ + S2- (weak electrolyte)
With decreased S2- ion concentration only Cu2+ is precipitated as CuS.
(b) (i) Schottky defect: It is a stoichiometric defect in which the equal number of cations and anions in the ionic crystal are missing from their lattice sites and electrical neutrality is maintained.
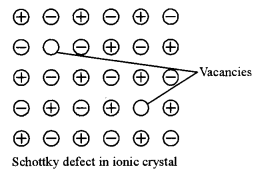

Question 4.
(a) (i) Give Lewis’definition of acids and bases. [1]
(ii) The solubility of Ag2CrO4 at 25°C is 8.0 × 10-5 moles litre-1. Calculate its solubility product. [1]
(b) (i) Define molar conductance of a solution. State its unit. How is it related to the specific conductance of a solution? [2]
(ii) Calculate the value of Ecell at 298 K for the following cell: [3]

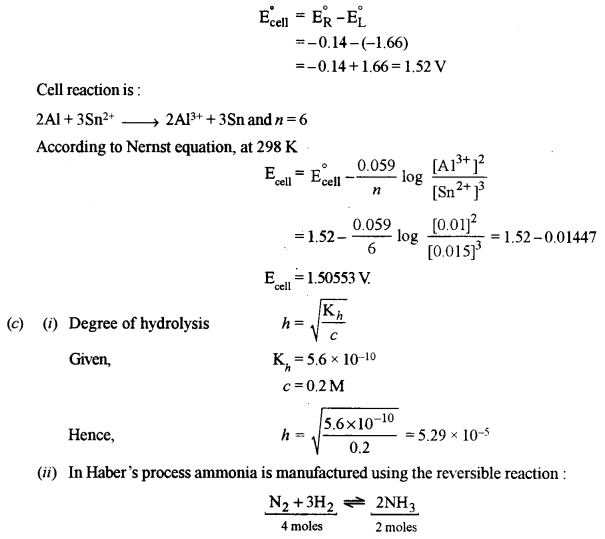
(c) (i) Calculate the degree of hydrolysis of 0.2 (M) sodium acetate solution. [1]
(Hydrolysis constant of sodium acetate = 5.6 × 10-10 and ionic product of H2O = 10-14 at 25°C)
(ii) Explain why high pressure is used in the manufacture of ammonia by Haber’s process. State the law or principle used. [2]
Answer:
(a) (i) A Lewis acid is defined as a substance (atom, ion or molecule) which is capable of accepting a pair of electrons.
Examples: Ag+, H+, AlCl3.
A Lewis base is defined as a substance (atom, ion or molecule) which is capable of donating a pair of electron.
Examples: NH3, Cl–.
(ii) Let the solubility of Ag2CrO4 in s mol L-1
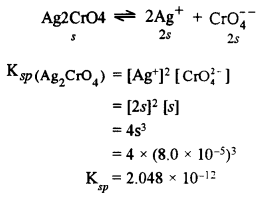
(b) (i) Molar conductance: It is defined as the conductance of all ions produced from one mole of an electrolyte dissolved in a definite volume of the solution.
Its unit is ohm-1 cm2 mol-1. Molar conductance ∧m is related to specific conductance k by the following equation:
\(\wedge_{m}=\frac{k \times 1000}{\mathrm{M}}\)
where k is specific conductance and M is a molar concentration of the solution.
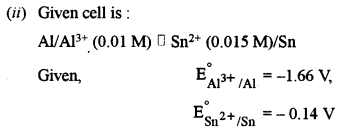
The forward reaction is accompanied by a decrease in the number of moles. On increasing pressure, then according to Le-Chatelier’s principle, the equilibrium will shift in a direction in which pressure decreases i.e., decrease in a number of moles takes place i.e., favour the formation of ammonia. Or in other words, an increase in pressure will favour the formation of ammonia. This is in accordance with Le-Chatelier’s principle, the law states ‘If a system at equilibrium is subjected to a change of concentration, temperature and pressure, then the equilibrium shifts itself in a direction that tends towards the effect of change imposed.’
Section – B
(Answer any two questions)
Question 5.
(a) Give the IUPAC names of the following coordination compounds: [2]
(i) K2[Zn(OH)4]
(ii) [CO(NH3)5 (CO3)] Cl.
(b) For the complex ion [Fe (CN)6]3- state: [1]
(i) The geometry of the ion.
(ii) The magnetic property of the ion.
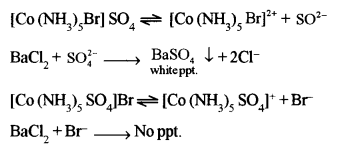
(c) What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers. [2]
Answer:
(a) (i) Potassium tetrahydroxozincate (II)
(ii) Pentaamminecarbonatocobalt (III) chloride
(b) (i) Octahedral geometry
(ii) Ion is paramagnetic.
(c) [Co(NH3)5 Br] SO4 and [Co(NH3)5SO4] Br are ionisation isomers.
The aqueous solution of [Co(NH3)5 Br] SO4 will give a white ppt. with BaCl2 solution insoluble in conc. HNO3, confirming the presence of free \(\mathrm{SO}_{4}^{2-}\) ion. An aqueous solution of [Co(NH3)5SO4] Br will not give this test.
Question 6.
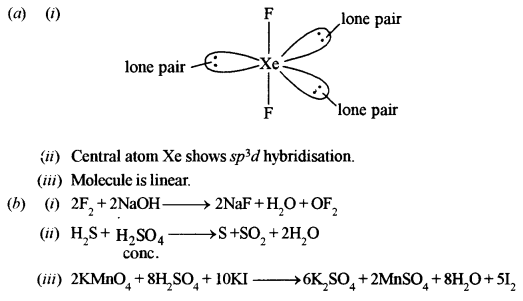
(a) For the molecule XeF2: [2]
(i) Draw the structure of the molecule indicating the lone pairs.
(ii) State the hybridisation of the central atom.
(iii) State the geometry of the molecule.
(b) Give balanced chemical equations for the following reactions: [2]
(i) Fluorine treated with dilute sodium hydroxide reactions.
(ii) Hydrogen sulphide treated with concentrated sulphuric acid.
(iii) Potassium iodide treated with acidified potassium permanganate solution.
Answer:
Question 7.
(a) In the extraction of zinc from zinc blende: [2]
(i) Give an equation to show how zinc oxide is converted to zinc.
(ii) How is impure zinc finally electro-refined?
(b) Explain why:
(i) Transition elements form coloured compounds. [2]
(ii) Interhalogen compounds are more reactive than their constituent elements.
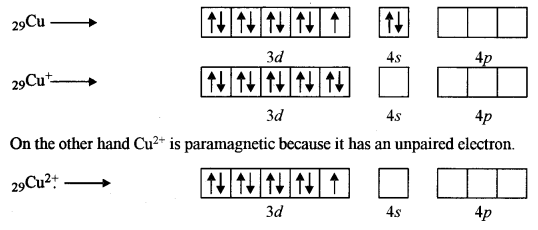
(iii) Cu+ is diamagnetic but Cu2+ is paramagnetic. (Z = 29)
Answer:
![]()
(ii) Electro-refining of zinc:
The electrolyte used: ZnSO4 solution containing little H2SO4.
Anode: Block of impure zinc.
Cathode: Thin sheet of pure zinc.
On passing current zinc is obtained at the cathode.
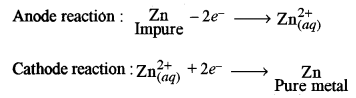
(b) (i) Transition metal ions generally possess one or more unpaired electrons. By absorbing a requisite amount of energy from visible light, the unpaired electrons present in the lower energy d-orbital get promoted to high energy d-orbitals. This is called a d-d transition. The energy involved in d-d transition is very small and such radiations are present within the visible region of light. Transition metal ions absorb such radiations from visible light and remaining wavelengths present in the visible region get transmitted and this imparts colour.
(ii) Interhalogen compounds are more reactive than their constituent elements because X-Y bonds present between two different halogens in them are weaker than X-X or Y-Y bonds.
(iii) Cu+ is diamagnetic because it has no impaired electrons in its core.
Section – C
(Answer any two questions)
Question 8.
(a) How can the following conversions be brought about: [3]
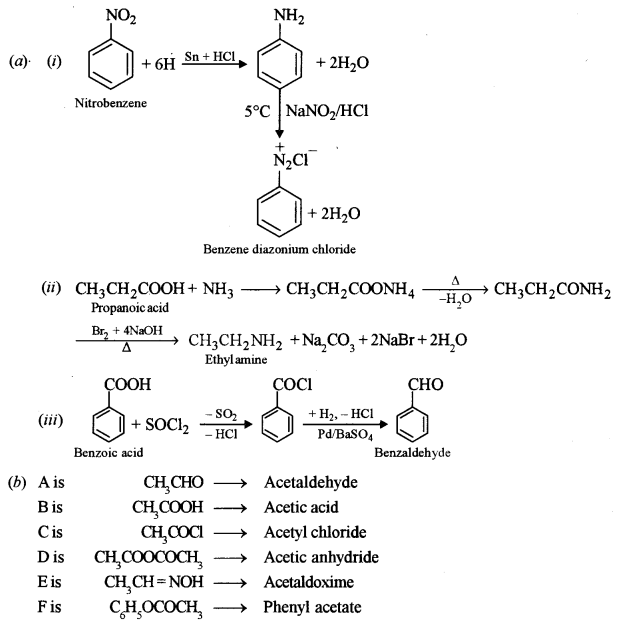
(i) Nitrobenzene to benzene diazonium chloride.
(ii) Propanoic acid to ethylamine.
(iii) Benzoic acid to benzaldehyde. [2]
(b) Identify the compounds A, B, C, D, E and F: [3]
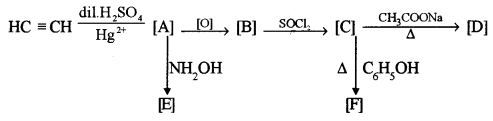
Answer:
Question 9.
(a) (i) Acetamide is heated with bromine and sodium hydroxide solution. [1]
(ii) Benzaldehyde is treated with 50% sodium hydroxide solution.
(b) Give one chemical test to distinguish between the following pairs of compound: [2]
(i) Acetone and phenol.
(ii) Formic acid and acetic acid.
(c) (i) Name the type of isomerism exhibited by the following pairs of compound: [2]
(1) (C2H5)2NH and CH3-NH-C3H7
(2) 1 – butanol and 2 methyl-1 -propanol.
(ii) Name the type of isomerism that the compound with molecular formula C3H6O2 exhibits. Represent the isomers.
Answer:
(a) (i) Methylamine is formed.
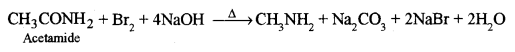
This is Hoffmann’s bromamide reaction.
(ii) Sodium benzoate and benzyl alcohol are formed.
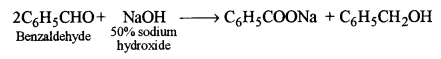
This is Cannizzaro’s reaction.
(b) (i) Acetone, when heated with NaOH/I2, gives a yellow crystal of iodoform.
![]()
Phenol does not give this test.
(ii) Formic acid gives a silver mirror with tollen’s reagent.
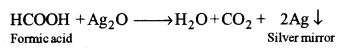
Acetic acid does not give this test.
Question 10.
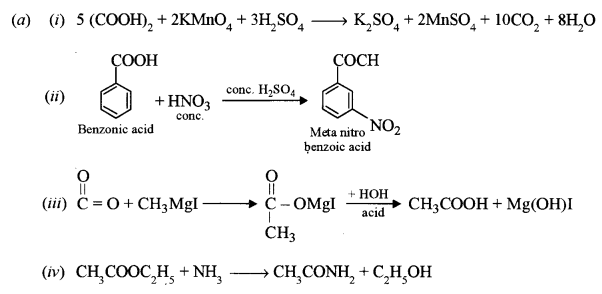
(a) Write balanced chemical equations for the following reactions: [4]
(i) Oxalic acid is treated with acidified potassium permanganate solution.
(ii) Benzoic acid is treated with a mixture of concentrated nitric acid and concentrated sulphuric acid.
(iii) Methyl magnesium iodide is treated with carbon dioxide and the product hydrolysed in acidic medium.
(iv) Ethylacetate is treated with ammonia.
(b) An organic compound [A] having molecular formula C2H7N on treatment with nitrous acid gives a compound [B] having molecular formula C2H6O. [B] on treatment with an organic compound [C] gives a carboxylic acid [D] and a sweet-smelling compound [E]. Oxidation of [B] with acidified potassium dichromate also gives [D]. [4]
(i) Identify [A], [B], [C], [D] and [E].
(ii) Write a balanced chemical equation of [D] with chlorine in the presence of red phosphorus and name the reaction.
(c) Acetamide is amphoteric in nature. Give two equations to support this statement. [2]
Answer: