ISC Chemistry Previous Year Question Paper 2012 Solved for Class 12
Maximum Marks: 70
Time allowed: 3 hours
- Answer all questions in Part I and six questions from Part II, choosing two questions from Section A, two from Section B and two from Section C.
- All working, including rough work, should be done on the same sheet as, and adjacent to, the rest of the answer.
- The intended marks for questions or parts of questions are given in brackets [ ].
- Balanced equations must be given wherever possible and diagrams where they are helpful.
- When solving numerical problems, all essential working must be shown.
- In working out problems use the following data:
Gas constant R = 1.987 cal deg-1 mol-1 = 8.314 JK-1 mol-1 = 0.0821 dm3 atm K-1 mol-1. 1L atm = 1 dm3 atm = 101.3 J.
1 Faraday = 96500 Coulombs, Avogadro’s number = 6.023 × 1023
Part – I
(Answer all questions)
Question 1.
(a) Fill in the blanks by choosing the appropriate word/words from those given in the brackets: [5]
(Raoult’s, Arrhenius, lateral, sodium, magnesium, negative, positive, non-ideal, ideal, iron, copper, van’t Hoff, s, p, ethanol, ethanoic acid, methanoic acid, methanol, propanoic acid.)
(i) For a spontaneous change to take place, the ΔS of the system should be …………… and ΔG of the system should be ……….
(ii) Hydrolysis of methyl propanoate gives ……….. and …………
(iii) Solutions which strictly obey ………… law is called …………. solutions.
(iv) π bonds are formed by the ………… overlap of ………… orbitals.
(v) Zinc can displace ……….. from CuSO4 solution, but cannot displace …………. from MgSO4 solution.
(b) Complete the following statements by selecting the correct alternative from the choices given: [5]
(i) The quantity of electricity required to deposit 1.15 g of sodium from molten NaCl (Na = 23, Cl = 35.5) is:
(1) IF
(2) 0.5 F
(3) 0.05 F
(4) 1.5 F
(ii) When acetic acid is reacted with calcium hydroxide and the product is distilled dry, the compound formed is:
(1) Calcium acetate
(2) Acetone
(3) Acetaldehyde
(4) Acetic anhydride
(iii) The [OH–] concentration of a weak base is given by:
(1) ckb
(2) \(\sqrt{c k_{b}}\)
(3) \(\sqrt{k_{b} / c}\)
(4) \(\sqrt{k_{b}}\)
(iv) In a plot of log k vs 1/T, the slope is
(1) -Ea/2.303
(2) Ea/2.303R
(3) Ea/2.303
(4) -Ea/2.303R
(v) Among the following coordination compounds, the one giving a white precipitate with BaCl2 solution is:
(1) [Cr(H2O)5Br]SO4
(2) [Cr(H2O)5SCN]
(3) [CO(NH3)5SO4] Br
(4) [Pt(NH3)6]Cl4
(c) Answer the following questions: [5]
(i) A solution X is prepared by dissolving three moles of glucose in one litre of water and a solution Y is prepared by dissolving 1.5 moles of sodium chloride in one litre of water. Will the osmotic pressure of X be higher, lower or equal to that of Y? Give a reason for your answer.
(ii) Give one example (equation) of a homogeneously catalysed reaction and name the catalyst.
(iii) Write the formula of the product formed when formaldehyde reacts with ammonia and name the product.
(iv) If the ionization (dissociation) constant of acetic acid is ka, what will be the pH of a solution containing equal concentrations of acetic acid and sodium acetate?
(v) What is the electronic configuration of the chromium atom (Z = 24)? Give a reason for your answer.
(d) Match the following: [5]
| (i) Nernst equation | (a) Water |
| (ii) Lactic acid | (b) Constant volume |
| (iii) Amphiprotic solvent | (c) Ammonia |
| (iv) Lewis base | (d) Optical isomers |
| (v) Isochoric process | (e) Electrochemical cells |
Answer:
(a) (ii) methanol, propanoic acid
(iii) Raoult’s, ideal
(v) copper, magnesium.
(b) (i) (3) 0.05 F
(ii) (2) Acetone
(iii) (3) \(\sqrt{k_{b} / c}\)
(iv) (4) -Ea/2.303R
(v) (1) [Cr(H2O)5Br]SO4
(c) (i) The osmotic pressure of X will be equal to that of Y.
Reason: Osmotic pressure is a colligative property. Its value depends upon the number of particles of solute present in the solution. 3 moles of glucose gives 3 moles of molecules (particles) in solution. 1.5 moles of NaCl, due to complete dissociation provides 3 moles of ions (particles) in solution.
Molar concentrations of solutions are the same and their osmotic pressures are also equal.
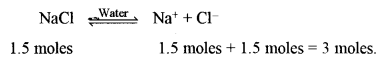
(ii) Hydrolysis of ethyl acetate catalysed by dil. H2SO4 to give ethanol and acetic acid.
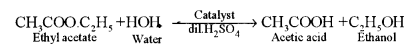
(iii) Urotropine or hexamethylenetetramine having formula (CH2)6N4 is formed.
(iv) For the equal concentration of acetic acid and sodium acetate in a solution, its pH will be given by
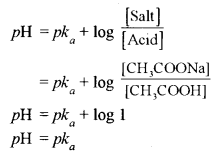
(v) Electronic configuration of Cr atom is 1s2, 2s22p6, 3s2p6d5, 4s1
This is because in this electronic configuration d subshell is half-filled which is more stable configuration than normally expected i.e., 3d44s2.
(d) (i) (e)
(ii) (d)
(iii) (a)
(iv) (c)
(v) (b)
Part – II
Answer six questions choosing two from Section A, two from Section B and two from Section C.
Section – A
(Answer any two questions)
Question 2.
(a) (i) A solution of urea in water has a boiling point of 100.18°C. Calculate the freezing point of the solution. (Kf for water is 1.86 K kg mol-1 and for water is 0.512 K kg mol-1). [2]
(ii) A solution of lactose-containing 8.45 g of lactose in 100 g of water has a vapour pressure of 4.559 mm of Hg at 0°C. If the vapour pressure of pure water is 4.579 mm of Hg, calculate the molecular weight of lactose. [3]
(b) (i) The molecular weight of H2S is more than that of H2O, but H2S is a gas and H2O is a liquid. Explain. [2]
(ii) When potassium cyanide reacts with water, will the resulting solution be acidic, alkaline or neutral? Justify your answer. [2]
(c) What is the hybridization of the carbon atom in ethyne molecule? What is the H-C-H bond angle? [1]
Answer:
(a) (i) Kb = 0.512 K kg mol-1
Kf = 1.86 K kg mol-1
ΔTb = 100.18° – 100° = 0.18
ΔT = Kb × molality
molality = 0.18/0.512
Now, ΔTf = Kf × molality
\(=1.86 \times \frac{0.18}{0.512}=0.6539\)
Freezing point of solutions = 0 – 0.6539 = -0.6539
(ii) Weight of lactose w = 8.45 g
Weight of water W = 100 g
Vapour pressure of pure water (p°) = 4.579 mm of Hg
Vapour pressure of solution (ps) = 4.559 mm of Hg
Now, according to Raoult’s’ law
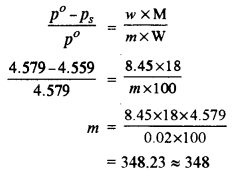
The molecular mass of lactose is 348.
(b) (ii) KCN is a salt of a strong base and weak acid. It undergoes hydrolysis when dissolved in water to form weak acid HCN and strong base KOH.
\(\begin{array}{c}{\mathrm{KCN}+\mathrm{HOH} \rightleftharpoons \mathrm{HCN}+\mathrm{KOH}} \\ {\mathrm{H}^{+}+\mathrm{CN}^{-} \rightleftharpoons \mathrm{HCN} \text { (anionic hydrolysis) }}\end{array}\)
HCN is a weak acid because it gives less H+ ions in the solution while KOH is strong electrolyte and remains completely dissociated. Therefore, the resulting solution contains more OH– ions and it is therefore alkaline.
Question 3.
(a) (i) State the second law of thermodynamics in terms of the entropy of the universe. [1]
(ii) Calculate the maximum work that can be obtained from the given electrochemical cell constructed with two metals M and N. [3 ]
\(\begin{array}{l}{\left[\mathrm{E}_{\mathrm{M}^{+}}^{o}+2_{/ \mathrm{M}}=-0.76 \mathrm{V}, \mathrm{E}_{\mathrm{N}^{+}+2 / \mathrm{N}}^{o}=0.34 \mathrm{V}\right]} \\ {\text { The cell reaction is } \mathrm{M}+\mathrm{N}^{+2} \rightarrow \mathrm{M}^{+2}+\mathrm{N}}\end{array}\)
(b) (i) To precipitate group III cations NH4Cl should be added to the solution before the addition of ammonium hydroxide. Explain why. [2]
(ii) A study of chemical kinetics of the reaction A + B → products, gave the following data at 25°C: [2]
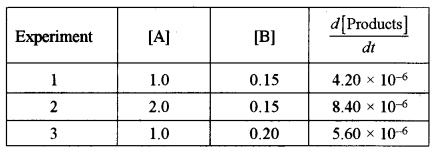
Find:
(1) The order of reaction with respect to A.
(2) The order of reaction with respect to B.
(3) The rate law.
(c) (i) What are F- centres in an ionic crystal?
(ii) Why are crystals having F-centres paramagnetic?
Answer:
(a) (ii) According to given cell reaction

(b) (i) To precipitate group III cations as hydroxides, a low cone, of OH– is required because solubility products of group III hydroxides are low as compared to the hydroxides of higher groups. Presence of NH4Cl which is a strong electrolyte suppresses dissociation of weak electrolyte NH4OH by common ion effect and decreases OH– ion concentration considerably.
\(\begin{array}{c}{\mathrm{NH}_{4} \mathrm{Cl} \rightleftharpoons \mathrm{NH}_{4}^{+}+\mathrm{Cl}^{-}} \\ {\mathrm{NH}_{4} \mathrm{OH} \rightleftharpoons \mathrm{NH}_{4}^{+}+\mathrm{OH}^{-}}\end{array}\)
(c) (i) F-centres: These are the anionic vacancies occupied by electrons. In ionic compounds having non-stoichiometric defects. These electrons remain delocalised and are responsible for imparting colours to crystals.
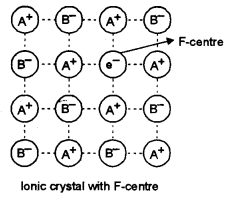
(ii) Due to presence of free impaired electrons occupying anionic vacancies a magnetic moment results by the spin of electron in a particular direction. The ionic crystals with F-centres, therefore, show paramagnetic behaviour.
Question 4.
(a) (i) The central atom of methane and water is in the same state of hybridization, but the shapes of the two molecules are different. Discuss. [2]
(ii) The conductivity of 0.2 M KCl solution is 3 × 10-2 ohm-1cm-1. Calculate its molar conductance. [1]
(b) (i) Draw the valence shell molecular orbital diagram of the oxygen molecule and predict its magnetic nature. [2]
(ii) Calculate the solubility of lead chloride in water, if its solubility product is 1.7 × 10-5. [2]
(Pb = 206, Cl = 35.5)
(c) For a crystal of diamond, state: [2]
(i) The hybridisation of the carbon atom.
(ii) The coordination number of each carbon atom.
(iii) The type of lattice in which it crystalises.
(iv) The number of carbon atoms presents per unit cell.
Answer:

(c) (i) sp3 hybridisation.
(ii) Coordination number is 4.
(iii) Type of lattice: Giant network type with the fee arrangement of C atoms.
(iv) The number of particles per unit cell is 4.
Section – B
(Answer any two questions)
Question 5.
(a) Write the formulae of the following coordination compounds : [2]
(i) potassiumtetracyanonickel (0)
(ii) triamminetrinitrocobalt (III)
(b) [CoF6]3- is a coordination complexion. [2]
(i) What is the oxidation number of cobalt in the complex?
(ii) How many unpaired electrons are there in the complex?
(iii) State the magnetic behaviour of the complex.
(iv) Give the IUPAC name of the complex.
(c) Draw the structural isomer of [Co(NH3)5NO2)Cl2 and name the type of isomerism. [1]
Answer:
(a) (i) K4[Ni(CN)4]
(ii) [Co(NH3)3(NO2)3]
(b) (i) [COF6]3-
x + (6 × -1) = -3
Oxidation number of Co, x = +3
(ii) 4 unpaired electrons.
(iii) The complexion is paramagnetic.
(iv) Hexafluorocobaltate (III) ion.
(c) Structural isomer → [Co(NH3)5Cl]Cl.NO2
Type of isomerism → Ionisation isomerism.
Question 6.
(a) Give the equations for the conversion of argentite (Ag2S) to metallic silver. [2]
(b) Give balanced equations for the following reactions: [3]
(i) Acidified potassium permanganate and oxalic acid.
(ii) Ozone and mercury.
(iii) The action of heat on a mixture of sodium chloride and concentrated sulphuric acid.
Answer:
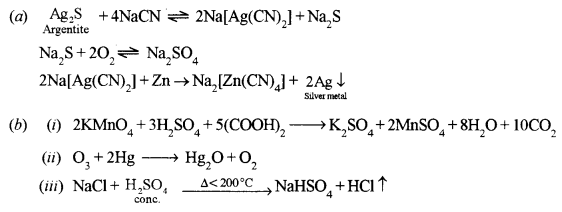
Question 7.
(a) Explain why transition metals form complex compounds. [2]
(b) (i) What is the hybridisation of the chlorine atom in the ClF3 molecule? [2]
(ii) Draw the structure of the molecule and state its geometry.
(c) Name the inert gases used for: [1]
(i) Filling sodium vapour lamps.
(ii) Obtaining light of different colours in neon signs.
Answer:
(a) Transition metals form complexes due to:
(i) Small size and high nuclear charge of these metals.
(ii) Availability of vacant d-orbitals of suitable energy to accommodate lone pairs of electrons donated by the ligands.
(b) (i) The hybridisation of the Cl atom in ClF3 is sp3d.
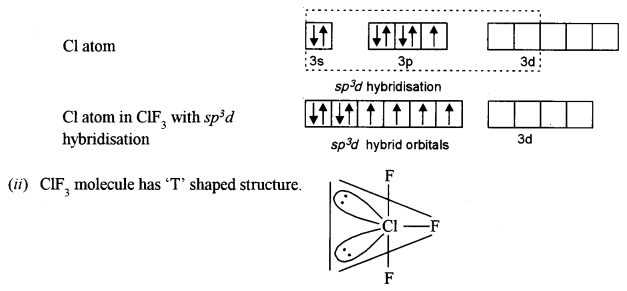
(c) (i) Neon gas
(ii) Argon mixed with neon gas.
Section – C
(Answer any two questions)
Question 8.
(a) How can the follow ing conversions be brought about:
(i) Ethanol to methylamine. [3]
(ii) Benzene to phenol. [2]
(b) Complete the following reactions and name the reactions: [3]
(i) ………… + 3I2 + 4KOH → CHI3 + CH3COOK + 3KI + 3H2O
(ii) C3H7NH2 + CHCl3 + 3KOH (alc.) → ……….. + 3KCl + 3H2O
(c) Name the type of polymerisation (addition or condensation) and name the monomers in each of the following polymers: [2]
(i) Protein.
(ii) Polyethene.
Answer:
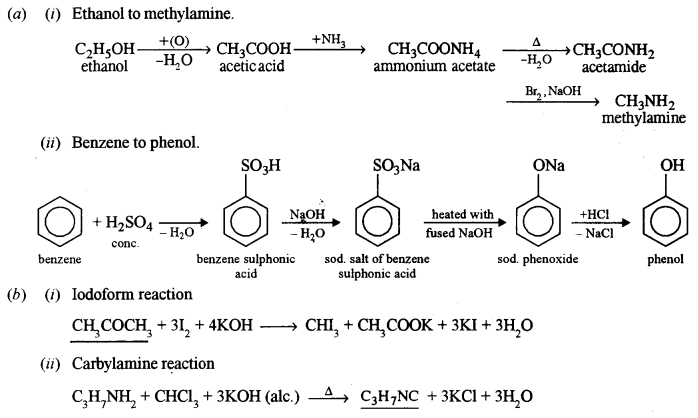
(c) (i) Condensation polymerisation,
monomer → amino acids.
(ii) Addition polymerisation,
monomer → ethylene.
Question 9.
(a) (i) What type of isomers are glucose and fructose? [1]
(ii) Name the functional group common to both glucose and fructose.
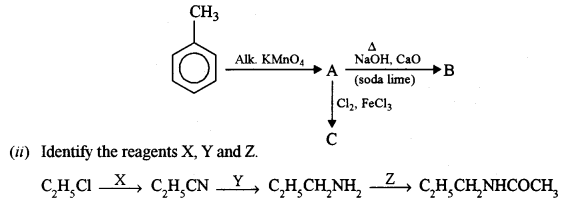
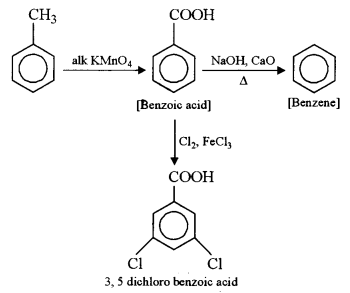
(b) (i) Identify the products A, B and C: [3]
(c) Give balanced equations for the following reactions: [3]
(i) Benzaldehyde and hydroxylamine.
(ii) Benzoic acid and phosphorus pentachloride.
(iii) 1-butanol and hydrogen chloride.
(d) Give one good chemical test to distinguish between the following pairs of compounds: [3]
(i) Methanal and ethanal.
(ii) Urea and benzoic acid.
Answer:
(a) (i) Functional isomers
(ii) Alcoholic group
(b) (i) A → Benzoic acid, B → Benzene, C → 3, 5 dichloro benzoic acid.
(ii) X → alcoholic KCN, Y → LiAlH4, Z → CH3COCl.
(c) (i) C6H5CHO + H2NOH → C6H5CH=NOH + H2O
(ii) C6H5COOH + PCl5 → C6H5COCl + POCl3 + HCl
(iii) CH3CH2CH2CH2OH + HCl → CH3CH2CH2CH2Cl + H2O
(d) (i) Ethanal, on warming with iodine and NaOH solution gives a yellow crystalline precipitate of iodoform. Methanal does not give this test.
(ii) Urea when heated just above its melting point and treated with alkali and a drop of dilute copper sulphate solution, gives violet colour. While benzoic acid does not give this test.
Question 10.
(a) An aliphatic hydrocarbon A on treatment with sulphuric acid in the presence of HgSO4 yields a liquid B with molecular formula C2H4O. B on oxidation with acidified potassium dichromate yields C which gives effervescence with sodium bicarbonate. C, when treated with SOCl2, gives D. WhenD reacts with ethanol it gives a sweet-smelling liquid E. E is also formed when C reacts with ethanol in the presence of cone. H2SO4. [4]
(i) Identify A, B, C, D and E.
(ii) Draw the structure of the isomer of compound B.
(iii) Write the balanced equation for the conversion of A to B.
(b) (i) The compound C6H12 shows optical isomerism. Draw the structural formula of the compound and name it. [1]
(ii) Name any three types of isomerisms that the compound with molecular formula C4H7Cl can give rise to. Also, represent the structures of the compounds relevant to these isomers. [3]
(c) Give equations to show what happens when a mixture of potassium cyanate and ammonium sulphate is strongly heated. Name the reaction. [2]
Answer:
(a) (i) A → Acetylene, B → Acetaldehyde, C → Acetic acid, D → Acetyl chloride, E → Ethyl acetate.
(ii) Isomers of acetaldehyde (CH2=CHOH)
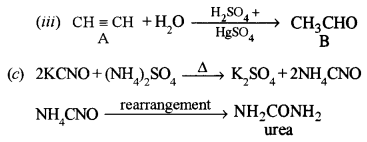
It is Wohler’s synthesis of urea.
