ICSE Chemistry Previous Year Question Paper 2015 Solved for Class 10
ICSE Paper 2015
CHEMISTRY
(Two Hours)
Answers to this Paper must be written on the paper provided separately.
You will not be allowed to write during the first 15 minutes.
This time is to be spent in reading the Question Paper.
The time given at the head of this Paper is the time allowed for writing the answers.
Section I is compulsory. Attempt any four questions from Section II.
The intended marks for questions or parts of questions are given in brackets [ ].
SECTION-I (40 Marks)
(Attempt all questions from this Section)
Question 1:
(a) Select from the list the gas that matches the description given in each case:
[ammonia, ethane, hydrogen chloride, hydrogen sulphide, ethyne]
- This gas is used as a reducing agent in reducing copper oxide to copper.
- This gas produces dense white fumes with ammonia gas.
- This gas is used for welding purposes.
- This gas is also a saturated hydrocarbon.
- This gas has a characteristic rotten egg smell. [5]
(b) Choose the most appropriate answer for each of the following:
- Among the elements given below, the element with the least electronegativity is:
(A) Lithium (B) Carbon
(C) Boron (D) Fluorine - Identify the statement which does not describe the property of alkenes:
(A) They are unsaturated hydrocarbons
(B) They decolourise bromine water
(C) They can undergo addition as well as substitution reactions
(D) They undergo combustion with oxygen forming carbon dioxide and water - This is not an alloy of copper:
(A) Brass (B) Bronze
(C) Solder (D) Duralumin - Bonding in this molecule can be understood to involve coordinate bonding.
(A) Carbon tetrachloride (B) Hydrogen
(C) Hydrogen chloride (D) Ammonium chloride - Which of the following would weigh the least?
(A) 2 gram atoms of Nitrogen.
(B) 1 mole of Silver
(C) 22.4 litres of oxygen gas at 1 atmospheric pressure and 273 K.
(D) 6.02 x 1023 atoms of carbon.
[Atomic masses: Ag = 108, N = 14, O = 16, C = 12] [5]
(c) Complete the following calculations. Show working for complete credit:
(i) Calculate the mass of Calcium that will contain the same number of atoms as are present in 3.2 gm of Sulphur.
[Atomic masses: S = 32, Ca = 40] [2]
(ii) If 6 litres of hydrogen and 4 litres of chlorine are mixed and exploded and if water is added to the gases formed, find the volume of the residual gas. [2]
(iii) If the empirical formula of a compound is CH and it has a vapour density of 13, find the molecular formula of the compound. [1]
(d) State one relevant observation for each of the following:
- When crystals of copper nitrate are heated in a test tube.
- When the gaseous product obtained by dehydration of ethyl alcohol is passed through bromine water.
- When hydrogen sulphide gas is passed through lead acetate solution.
- When ammonia gas is burnt in an atmosphere of excess oxygen.
- At the Anode when aqueous copper sulphate solution is electrolysed using copper electrodes. [5]
(e) Identify the acid which matches the following description (i) to (v):
- The acid which is used in the preparation of a non-volatile acid.
- The acid which produces sugar charcoal from sugar.
- The acid which is prepared by catalytic oxidation of ammonia.
- The acid on mixing with lead nitrate solution produces a white precipitate which is insoluble even on heating.
- The acid on mixing with silver nitrate solution produces a white precipitate which is soluble in excess ammonium hydroxide. [5]
(f) Give appropriate scientific reasons for the following statements:
- Zinc oxide can be reduced to zinc by using carbon monoxide, but aluminium oxide cannot be reduced by a reducing agent.
- Carbon tetrachloride does not conduct electricity.
- During electrolysis of molten lead bromide graphite anode is preferred to other electrodes.
- The electrical conductivity of acetic acid is less in comparison to the electrical conductivity of dilute sulphuric acid at a given concentration.
- Electrolysis of molten lead bromide is considered to be a redox reaction. [5]
(g) (i) Give balanced chemical equations for the following conversions A, B and C: [3]
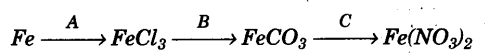
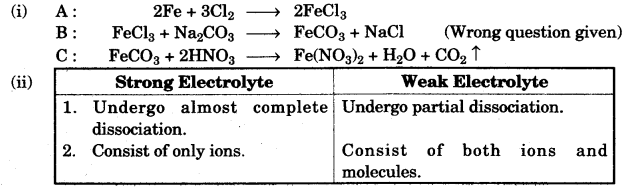
(ii) Differentiate between the terms strong electrolyte and weak electrolyte. (stating any two differences) [2]
(h) Answer the following questions:
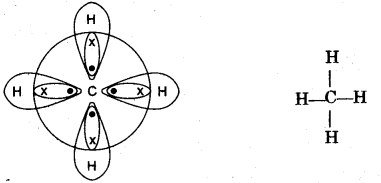
(i) Explain the bonding in methane molecule using electron dot structure. [2]
(ii) The metals of Group 2 from top to bottom are Be, Mg, Ca, Sr and Ba.
- Which one of these elements will form ions most readily and why?
- State the common feature in the electronic configuration of all these elements. [3]
Answer:
(a)
- Ammonia
- Hydrogen Chloride
- Ethyne
- Ethane
- Hydrogen Sulphide
(b)
- (A) Lithium
- (C) They can undergo addition as well as substitution
- (C) Solder
- (D) Ammonium chloride
- (D) 6.02 x 1023 atoms of carbon
(c)

(d)
- Release reddish brown gas NO2 (Nitrogen-di-oxide) which turns freshly prepared Ferrous Sulphate solution brown.
The residue left is black copper oxide. - Ethyl alcohol on dehydration forms ethene which when passed through bromine water makes it colourless.
- The yellow lead acetate paper turns greyish black with hydrogen sulphide gas due to formation of lead (II) Sulphide.
- Ammonia bums with a green flame.
- Nothing is left at anode as copper atoms form copper ions and migrate towards cathode.
(e)
- Conc. Sulphuric acid
- Conc. Sulphuric acid
- Nitric acid
- Sulphuric acid
- Hydrochloric acid
(f)
- Zinc is comparatively less reactive. So its oxideis less stable and hence it can be reduced by reducing agent whereas Aluminium is very reactive due to which its oxide is quite stable and it can not be reduced by a reducing agent it can be reduced only by electrolytic reduction.
- Carbon tetrachloride is a non polar covalent compound and so do not have free ions therefore they do not conduct electricity.
- Graphite is an inert electrode which is preferred in electrolysis of molten lead bromide because if other active electrodes are used then bromine will react with them.
- Acetic acid is a weak acid and has less number ions so conductivity is less whereas dil. sulphuric acid is a strong acid, has more number of ions and thererfore its electrical conductivity is more.
- During electrolysis of molten lead bromide lead gets oxidised and bromine gets reduced. Thus both oxidation and reduction occur simultaneously. So it is a redox reaction.
(g)
(h)
(i) Methane: CH4
C = 6 = 2, 4
H = 1
In bonding of methane molecule, one atom of carbon shares four electron pairs, one with each of the four atoms of hydrogen.
(ii)
- Barium will form ions most readily because it is most metallic and lose electrons readily and form positive ion.
- All these elements have 2 valence electrons.
SECTION-II (40 Marks)
(Answer any four questions from this section)
Question 2:
(a) Arrange the following as per the instructions given in the brackets:
- Cs, Na, Li, K, Rb (increasing order of metallic character)
- Mg, Cl, Na, S, Si (decreasing order of atomic size)
- Na, K, Cl, S, Si (increasing order of ionization energy)
- Cl, F, Br, I (increasing order of electron affinity) [4]
(b) Choose the most appropriate answer from the following list of oxides which fit the description. Each answer may he used only once:
[SO2, SiO2 Al2O3, MgO, CO, Na2O]
- A basic oxide.
- An oxide which dissolves in water forming an acid.
- An amphoteric oxide
- A covalent oxide of a metalloid. [4]
(c) Element X is a metal with a valency 2, Y is a non-metal with a valency 3.
- Write an equation to show how Y forms an ion.
- If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound. [2]
Answer:
(a)
- Li < Na < K < Rb < Cs
- Na > Mg > Si > S > Cl
- K < Na < Si < S < Cl
- I < Br < F < Cl
(b)
- MgO
- SO2
- Al2O3
- CO
(c)
- Y + 3e– → Y– – –
- 3X + Y2 → X3Y2
Question 3:
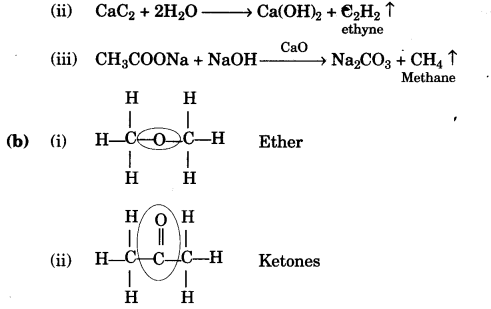
(a) Give balanced chemical equations for the following conversions:
(i) Ethanoic acid to ethyl ethanoate.
(ii) Calcium carbide to ethyne.
(iii) Sodium ethanoate to methane. [3]
(b) Using their structural formulae identify the functional group by circling them:
(i) Dimethyl ether.
(ii) Propanone. [2]
(c) Name the following:
- Process by which ethane is obtained from ethene.
- A hydrocarbon which contributes towards the greenhouse effect.
- Distinctive reaction that takes place when ethanol is treated with acetic acid.
- The property of elements by virtue of which atoms of the element can link to each other in the form of a long chain or ring structure.
- Reaction when an alkyl halide is treated with alcoholic potassium hydroxide. [5]
Answer:
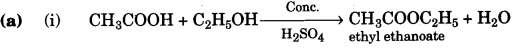
(c)
- Hydrogenation
- Methane
- Esterification
- Catenation
- Dehydrohalogenation
Question 4:
(a) Identify the anion present in each of the following compounds:
- A salt M on treatment with concentrated sulphuric acid produces a gas which fumes in moist air and gives dense fumes with ammonia.
- A salt D on treatment with dilute sulphuric acid produces a gas which turns lime water milky but has no effect on acidified potassium dichromate solution.
- When barium chloride solution is added to salt solution E a white precipitate insoluble in dilute hydrochloric acid is obtained. [3]
(b) The following table shows the tests a student performed on four different aqueous solution which are X, Y, Z and W. Based on the observations provided, identify the cation present: [4]
| Chemical test | Observation | Conclusion |
| To solution X, ammonium hydroxide is added in minimum quantity first and then in excesss. | A dirty white precipitate is formed which dissolves in excess to form a clear solution. | (i) |
| To solution Y, ammonium hydroxide is added in minimum quantity first and then in excess. | A pale blue precipitate is formed which dissolves in excess to form a clear inky blue solution. | (ii) |
| To solution W, a small quantity of sodium hydroxide solution is added and then in excess. | A white precipitate is formed which remains insoluble. | (iii) |
| To a salt Z, calcium hydroxide solution is added and then heated. | A pungent smelling gas turning moist red litmus paper blue is obtained. | (iv) |
(c) Give balanced chemical equations for each of the following:
- Lab preparation of ammonia using an ammonium salt.
- Reaction of ammonia with excess chlorine.
- Reaction of ammonia with sulphuric acid. [3]
Answer:
(a)
- M = Chloride (Cl–)
- D = Carbonate (CO3– – )
- E = Sulphate (SO4– –)
(b)
- Zn++ = Zinc ion.
- Cu++ = Copper ion.
- Ca++ = Calcium ion.
- NH4+ = Ammonium ion.
(c)
- 2NH4Cl + Ca(OH)2 → CaCl2 + 2NH3 + 2H2O
- NH3 + 3Cl2 → NCl3 + 3HCl
- 2NH3 + H2SO4 → (NH4)2SO4
Question 5:
(a) Consider the following reaction and based on the reaction answer the questions that follow:
![]()
Calculate:
(i) the quantity in moles of (NH4)2Cr2O7 if 63 gm of (NH4)2Cr2O7 is heated. [1]
(ii) the quantity in moles of nitrogen formed. [1]
(iii) the volume in litres or dm3 of N2 evolved at S.T.P. [1]
(iv) the mass of grants of Cr2O3 formed at the same time. [2]
[Atomic masses: H = 1, Cr = 52, N = 14]
(b) (i) For each of the substance listed below, describe the role played in the extraction of aluminium:
- Cryolite
- Sodium hydroxide
- Graphite. [3]
(ii) Explain why:
- In the electrolysis of alumina using the Hall Heroult’s Process the electrolyte is covered with powdered coke.
- Iron sheets are coated with zinc during galvanisation. [2]
Answer:
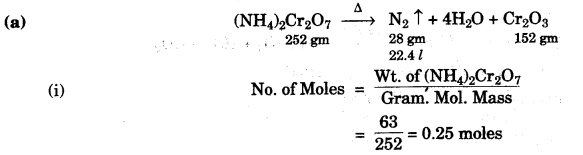

(b) (i)
- Cryolite: To reduce the melting point of Alumina and increase the conductivity of solution.
- Sodium hydroxide: for purification of bauxite ore as a leaching agent.
- Graphite: As anode and make the reduction of Al2O3 easier.
(ii)
- Electrolyte is covered with powdered coke to prevent heat loss from the molten electrolyte.
- Iron sheets are coated with zinc by galvanisation to prevent rusting of Iron sheets.
Question 6:
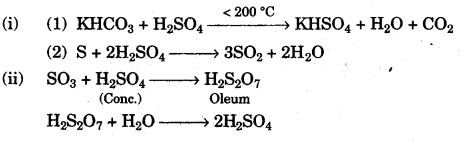
(a) (i) Give balanced chemical equations for the action of sulphuric acid on each of the following:
- Potassium hydrogen carbonate.
- Sulphur. [2]
(ii) In the contact process for the manufacture of sulphuric acid give the equations for the conversion of sulphur trioxide to sulphuric acid. [2]
(b) (i) Copy and complete the following table: [2]
| Anode | Electrolyte | |
| Purification of copper |
(ii) Write the equation taking place at the anode. [1]
(c) Explain the following:
- Dilute nitric acid is generally considered a typical acid but not so in its reaction with metals.
- Concentrated nitric acid appears yellow when it is left standing in a glass bottle.
- An all glass apparatus is used in the laboratory preparation of nitric acid. [3]
Answer:
(a)
(b)
(i)
| Anode | Electrolyte | |
| Purification of copper | Impure Copper | Aqueous solution of copper sulphate with few drops of sulphuric acid. |
(ii) At Anode: Cu – 2e– → Cu++
(c)
- Unlike other dilute acids only very dilute nitric acid (1%) reacts exclusively with Magnesium or Manganese to release hydrogen gas.
- Cone, nitric acid undergoes decomposition as it is very unstable to heat. On decomposition they form reddish brown nitrogen-di-oxide gas which when dissolved in the acid appears yellow in colour.
- Nitric acid is highly corossive and can damage plastic, rubber, wood, metal etc. therefore an all glass apparatus must be used.
Question 7:
(a) The following questions are pertaining to the laboratory preparation of hydrogen chloride gas:
(i) Write the equation for its preparation mentioning the condition required. [1]
(ii) Name the drying agent used and justify your choice. [2]
(iii) State a safety precaution you would take during the preparation of hydrochloric acid. [1]
(b) An element L consists of molecules:
- What type of bonding is present in the particles that make up L?
- When L is heated with iron metal, it forms a compound FeL. What chemical term would you use to describe the change undergone by L? [2]
(c) From the list of the following salts choose the salt that most appropriately fits the description given in the following:
[AgCl, MgCl2, NaHSO4, PbCO3, ZnCO3, KNO3, Ca(N03)2]
- A deliquescent salt.
- An insoluble chloride.
- On heating, this salt gives a yellow residue when hot and white when cold.
- On heating this salt, a brown coloured gas is evolved. [4]
Answer:
(a)
![]()
(ii) Drying agent is cone, sulphuric acid because it is an acidic drying agent and other drying agent react with HCl.
(iii) The temperature should be raised above 200°C during the preparation of hydrochloric acid because above this temperature it forms sticky substance sodium sulphate that sticks to glass apparatus which becomes difficult to remove and also cause wastage of heat.
(b)
- Covalent bonding.
- Reduction.
(c)
- A deliquescent salt — MgCl2
- An insoluble chloride — AgCl
- ZnCO3
- Ca(N03)2