ICSE Chemistry Previous Year Question Paper 2011 Solved for Class 10
ICSE Paper 2011
CHEMISTRY
(Two Hours)
Answers to this Paper must be written on the paper provided separately.
You will not be allowed to write during the first 15 minutes.
This time is to be spent in reading the Question Paper.
The time given at the head of this Paper is the time allowed for writing the answers.
Section I is compulsory. Attempt any four questions from Section II.
The intended marks for questions or parts of questions are given in brackets [ ].
SECTION-I (40 Marks)
(Attempt all questions from this Section)
Question 1:
(a) Choose from the following list of substances, as to what matches the description from (i) to (v) given below:
[Acetylene gas, aqua fortis, coke, brass, barium chloride, bronze, platinum].
- An aqueous salt solution used for testing sulphate radical.
- A catalyst used in the manufacture of nitric acid by Ostwald’s process.
- A black powdery substance used for the reduction of zinc oxide during its extraction.
- A gaseous hydrocarbon commonly used for welding purposes.
- The substance is an alloy of zinc, copper and tin. [5]
(b) What would you observe in each of the following cases?
- Ammonium hydroxide is first added in a small quantity and then in excess to a solution of copper sulphate.
- Sugar crystals are added to a hard glass test tube containing concentrated sulphuric acid.
- Copper is heated with concentrated nitric acid in a hard glass test tube.
- Water is added to the product formed, when aluminium is burnt in a jar of nitrogen gas.
- When carbon monoxide is passed over heated copper oxide. [5]
(c) Give reasons as to why:
- The electrolysis of acidulated water is considered to be an example of catalysis.
- Almost 90% of all known compounds are organic in nature.
- It is dangerous to burn methane in an insufficient supply of air.
- Hydrogen chloride can be termed as a polar covalent compound.
- The oxidising power of elements increases on moving from left to right
along a period in the periodic table. [5]
(d) Fill in the blanks from the choices given below: [5]
- In covalent compounds, the bond is formed due to the ……….. (sharing /transfer) of electrons.
- Electrovalent compounds have a …………. (low/high) boiling point.
- A molecule of …………. contains a triple bond (hydrogen, ammonia, nitrogen).
- Across a period, the ionization potential …………… (increases, decreases, remains same).
- Down the group, electron affinity ……………. (increases, decreases, remains same). [5]
(e)
(i) Calculate the volume of 320 g of SO2 at stp. (Atomic mass: S = 32 and O = 16).
(ii) State Gay-Lussac’s Law of combining volumes.
(iii) Calculate the volume of oxygen required for the complete combustion of 8.8 g of propane (C3H8). (Atomic mass: C = 14, O = 16, H = 1, Molar Volume 22.4 dm3 at stp). [5]
(f) Choose the correct answer from the options given below: [10]
- This metal is a liquid at room temperature.
(A) Potassium (B) Zinc
(C) Gold (D) Mercury - Hydroxide of this metal is soluble in sodium hydroxide solution:
(A) Magnesium (B) Lead
(C) Silver (D) Copper - In the periodic table alkali metals are plowed in the group ……..
(A) 1 (B) 11
(C) 17 (D) 18 - Hydrogen chloride gas being highly soluble in water is dried by:
(A) Anhydrous calcium chloride (B) Phosphorous penta oxide
(C) Quick lime (D) Concentrated sulphuric acid - The brown ring test is used for detection of:
(A) CO32- (B) NO3–
(O SO32- (D) Cl– - When dilute sulphuric acid reacts with iron sulphide, the gas evolved is ……….
(A) Hydrogen sulphide (B) Sulphur dioxide
(C) Sulphur trioxide (D) Vapour of sulphuric acid - The functional group present in acetic acid is:
(A) Ketonic C = 0 (B) Hydroxyl —OH
(C) Aldehydic —CHO (D) Carboxyl —COOH - The unsaturated hydrocarbons undergo:
(A) a substitution reaction (B) an oxidation reaction
(C) an addition reaction (D) none of the above - The number of C-H bonds in ethane molecule are:
(A) Four (B) Six
(C) Eight (D) Ten - Which of the following properties do not match with elements of the halogen family?
(A) They have seven electrons in their valence shell.
(B) They are highly reactive chemically.
(C) They are metallic in nature.
(D) They are diatomic in their molecular form.
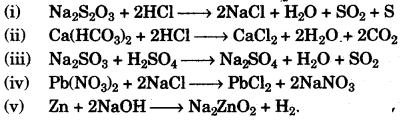
(g) Write the balanced chemical equation for each of the following reactions: [5]
(i) Sodium thiosulphate is reacted with dilute hydrochloric acid.
(ii) Calcium bicarbonate reacts with dilute hydrochloric acid.
(iii) Dilute sulphuric acid is poured over sodium sulphite.
(iv) Lead nitrate solution is added to sodium chloride solution.
(v) Zinc is heated with sodium hydroxide solution.
Answer:
(a)
- Barium chloride
- Platinum
- Coke
- Acetylene gas
- Bronze
(b)
- In small quantity — a bluish white ppt is obtained.
In excess — deep (inky) blue solution is obtained. - Turns to black mass called sugar charcoal.
- A reddish brown acidic gas called nitrogen dioxide is obtained.
- Pungent smelling basic gas called ammonia is evolved.
- Reddish metal copper is obtained.
(c)
- Sulphuric acid catalyses the ionisation of water because distilled water is a poor conductor of electricity and does not form ions.
- They all contains carbon as an essential component.
- It leads to incomplete combustion releasing carbon monoxide which is extremely poisonous and cause death.
- Difference between electronegativities of H and Cl is high. E.N. of Cl is much higher than that E.N. of H. So a unidirectional pull towards Cl will be created leading to polarity.
- Oxidising power means tendency to make the other element get oxidised which increases with increase in non metallic nature. From left to right in a period the non metallic nature increases.
(d)
- sharing
- high
- nitrogen
- increases
- decreases
(e)

(f)
- (D)
- (B)
- (A)
- (D)
- (B)
- (A)
- (D)
- (C)
- (B)
- (C)
(g)
SECTION-II (40 marks)
(Answer any four questions from this section)
Question 2:
(a) Differentiate between electrical conductivity of copper sulphate solution and copper metal. [3]
(b) Sodium hydroxide solution is added to the solutions containing the ions mentioned in List X. List Y gives the details of the precipitate. Match the ions with their coloured precipitates. [3]
| List X | List Y |
| 1. Pb2+ | A. Reddish brown |
| 2. Fe2+ | B. White insoluble in excess |
| 3. Zn2+ | C. Dirty green |
| 4. Fe3+ | D. White soluble in excess |
| 5. Cu2+ | E. White soluble in excess |
| 6. Ca2+ | F. Blue |
(c) During the electrolysis of copper (II) sulphate solution using platinum as cathode and carbon as anode:
(i) What do you observe at the cathode and at the anode? [1]
(ii) What change is noticed in the electrolyte? [1]
(iii) Write the reactions at the cathode and at the anode. [2]
Answer:
(a) During electrical conductivity of copper sulphate solution, the CuSO4 splits into Cu++ and SO4– – ions. Where as copper metal only conducts electricity but does not undergo any change.
(b)
| List X | List Y |
| 1. Pb2+ | D. White soluble in excess |
| 2. Fe2+ | C. Dirty green |
| 3. Zn2+ | E. White soluble in excess |
| 4. Fe3+ | A. Reddish brown |
| 5. Cu2+ | F. Blue |
| 6. Ca2+ | B. White insoluble in excess |
(c)
(i) Red metal copper deposits at cathode and colourless odourless gas oxygen at Anode.
(ii) The blue colour of copper sulphate solution soon fades.
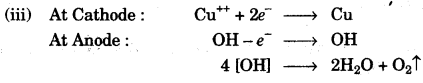
Question 3:
(a) Answer the following questions:
(i) Name a metal which is found abundantly in the earth’s crust.
(ii) What is the difference between calcination and roasting?
(iii) Name the process used for the enrichment of sulphide ore.
(iv) Write the chemical formulae of one main ore of iron and aluminium.
(v) Write the constituents of electrolyte for the extraction of aluminium. [5]
(b) The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
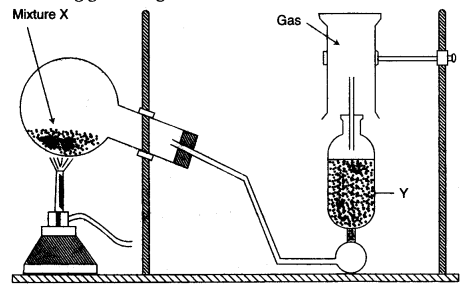
(i) Name the gas collected in the jar.
(ii) Write the balanced equation for the above preparation.
(iii) How is the gas being collected?
(iv) Name the drying agent used.
(v) How will you find that the jar is full of gas? [5]
Answer:
(a)

(b)
(i) Ammonia
![]()
(iii) Downward displacement of air.
(iv) Quick lime/CaO.
(v) Bring a moist red litmus paper to the mouth of the inverted jar if it immediately turns blue.
Or
Bring a glass rod dipped in hydrochloric acid to the mouth of the inverted jar, if it produces dense white fumes. The gas jar is full of gas.
Question 4:
(a) An organic compound with vapour density = 94 contains
C = 12.67%, H = 2.13% and Br = 85.11%. Find the molecular formula.
[Atomic mass: C = 12, H = 1, Br = 80] [3]
(b) Calculate the mass of:
(i) 1022 atoms of sulphur.
(ii) 0.1 mole of carbon dioxide.
[Atomic mass: S = 32, C = 12 and O = 16 and Avogadro’s Number = 6 × 1023] [2]
(c) In the laboratory preparation of hydrochloric acid, HCl gas is dissolved in water.
(i) Draw a diagram to show the arrangement used for the absorption of HCl in water.
(ii) Why is such an arrangement necessary? Give two reasons.
(iii) Write the chemical equations for the laboratory preparation of HCl gas when the reactants are:
(A) below 200 °C (B) above 200 °C. [5]
Answer:
(a)

(b)

(c)
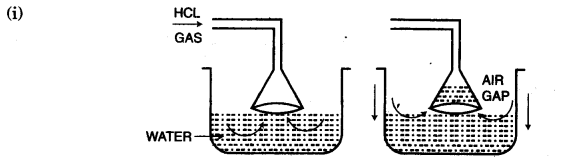
(ii) HCl gas is highly soluble in water almost at the ratio of 1 : 450. So HCl gas undergoes back suction due to which the source of HCl will be affected.
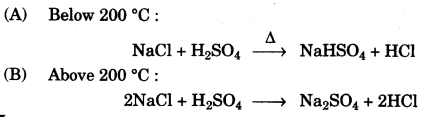
Question 5:
(a) Choose the correct word I phrase from within the brackets to complete the following sentences:
(i) The catalyst used for conversion of ethene to ethane is commonly …………… (nickel / iron / cobalt)
(ii) When acetaldehyde is oxidized with acidified potassium dichromate, it forms ………….. (ester / ethanol / acetic acid)
(iii) Ethanoic acid reacts with ethanol in presence of concentrated H2SO4 so as to form a compound and water. The chemical reaction which takes place is called …………… (dehydration/hydrogenation/esterification)
(iv) Write the equation for the reaction taking place between 1, 2-dibromethane and alcoholic potassium hydroxide.
(v) The product formed when ethene gas reacts with water in the presence of sulphuric acid is ………….. (ethanol / ethanal / ethanoic acid) [5]
(b) Write balanced chemical equations for the following:
(i) Monochloro ethane is hydrolysed with aqueous KOH.
(ii) A mixture of sodalime and sodium acetate is heated.
(iii) Ethanol under high pressure and low temperature is treated with acidified potassium dichromate.
(iv) Water is added to calcium carbide.
(v) Ethanol reacts with sodium at room temperature. [5]
Answer:
(a)
(i) Nickel
(ii) Acetic acid
(iii) Esterification
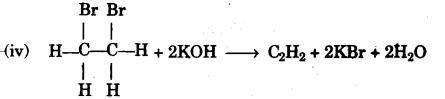
(v) Ethanol.
(b)
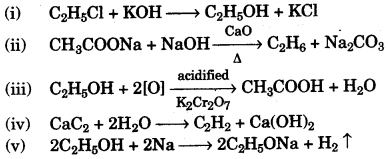
Question 6:
(a) (i) With the help of equations, given an outline for the manufacture of sulphuric acid by the contact process.
(ii) What property of sulphuric acid is shown by the reaction of concentrated sulphuric acid when heated with:
(A) potassium nitrate (B) carbon [5]
(b) (i) What is the special feature of the apparatus that is used in the laboratory preparation of nitric acid?
(ii) Why should the temperature of the reaction mixture of nitric acid not be allowed to rise above 200 °C? [2]
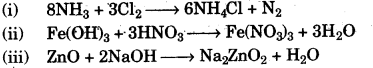
(c) Write balanced chemical equations for the following:
(i) Chlorine reacts with excess of ammonia.
(ii) Ferric hydroxide reacts with nitric acid.
(iii) Zinc oxide dissolves in sodium hydroxide. [5]
Answer:
(a)

(b)
(i) An all glass apparatus is used because nitric acid is highly corosive in nature and will damage wood, metal, rubber, plastic etc.
(ii)
- Above 200 °C the nitric acid decomposes into H2O, NO2 and O2 as it is very unstable to heat.
- Sodium sulphate or potassium sulphate will be formed which forms a crust over the mixture thus restricting the release of nitric acid vapours.
(c)
Question 7:
(a) (i) Give the number of the group and the period, of the element having three shells with three electrons in valence shell.
(ii) By drawing an electron dot diagram, show the lone pair effect leading to the formation of ammonium ion from ammonium gas and hydrogen ion.
(iii) What happens to the crystals of washing soda when exposed to air? Name the phenomenon exhibited. [5]
(b) Name the method used for preparation of the following salts from the list given below: [5]
(i) Sodium nitrate (ii) Iron (III) chloride.
(iii) Lead chloride (iv) Zinc sulphate (v) Sodium hydrogen sulphate.
List:
(A) Simple displacement (B) Neutralization
(C) Decomposition by acid (D) Double decomposition (E) Direct synthesis
Answer:
(a)
(i) Period number = 3
Group number = 13/III A.
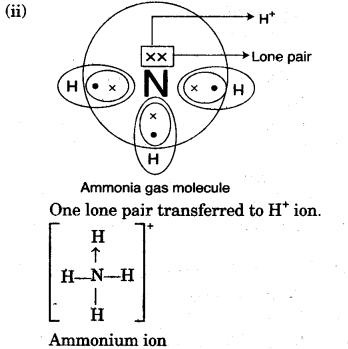
(iii) The crystals of washing soda when exposed to air, it loses its water of crystallisation crystals crumbles down to form white powder.
The phenomenon is called Efflorescence.
(b)
(i) Sodium nitrate — (B) Neutralisation.
(ii) Iron (III) chloride — (E) Direct Synthesis.
(iii) Lead chloride — (D) Double decomposition
(iv) Zinc sulphate — (A) Simple displacement.
(v) Sodium hydrogen sulphate — (C) Decomposition by acid.