Selina Concise Physics Class 10 ICSE Solutions Calorimetry APlusTopper.com provides step by step solutions for Selina Concise ICSE Solutions for Class 10 Physics Chapter 11 Calorimetry. You can download the Selina Concise Physics ICSE Solutions for Class 10 with Free PDF download option. Selina Publishers Concise Physics for Class 10 ICSE Solutions all questions are solved and … [Read more...] about Selina Concise Physics Class 10 ICSE Solutions Calorimetry
ICSE
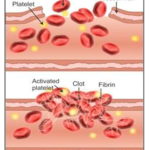
Selina Concise Biology Class 10 ICSE Solutions The Circulatory System
Selina Concise Biology Class 10 ICSE Solutions The Circulatory System APlusTopper.com provides step by step solutions for Selina Concise ICSE Solutions for Class 10 Biology Chapter 7 The Circulatory System. You can download the Selina Concise Biology ICSE Solutions for Class 10 with Free PDF download option. Selina Publishers Concise Biology for Class 10 ICSE Solutions all … [Read more...] about Selina Concise Biology Class 10 ICSE Solutions The Circulatory System
Selina ICSE Solutions for Class 10 Maths – Compound Interest (Using Formula)
Selina ICSE Solutions for Class 10 Maths - Compound Interest (Using Formula) Selina ICSE Solutions for Class 10 Maths Chapter 2 Compound Interest (Using Formula)Exercise 2(A)Solution 1: Given : P = ₹ 12,000; n = 3 years and r = 5%= ₹ 13,891.50 C.I. = ₹ 13,891.50 - ₹ 12,000 = ₹ 1,891.50Solution 2: Given : P = ₹ 15,000; n = 2 years ; r1 = 8%; r2 = 10%= ₹ … [Read more...] about Selina ICSE Solutions for Class 10 Maths – Compound Interest (Using Formula)
Selina Concise Mathematics Class 10 ICSE Solutions Chapter 1 GST (Goods and Services Tax)
Selina Concise Mathematics Class 10 ICSE Solutions Chapter 1 GST (Goods and Services Tax) Students should practice Goods & Service Tax (GST) – CS Executive Tax Laws MCQ Questions with Answers based on the latest syllabus.Selina Publishers Concise Mathematics Class 10 ICSE Solutions Chapter 1 GST (Goods and Services Tax) GST Exercise 1A - Selina Concise Mathematics … [Read more...] about Selina Concise Mathematics Class 10 ICSE Solutions Chapter 1 GST (Goods and Services Tax)
Selina Concise Mathematics Class 9 ICSE Solutions Compound Interest (Using Formula)
Selina Concise Mathematics Class 9 ICSE Solutions Compound Interest (Using Formula) APlusTopper.com provides step by step solutions for Selina Concise Mathematics Class 9 ICSE Solutions Chapter 3 Compound Interest (Using Formula). You can download the Selina Concise Mathematics ICSE Solutions for Class 9 with Free PDF download option. Selina Publishers Concise Mathematics for … [Read more...] about Selina Concise Mathematics Class 9 ICSE Solutions Compound Interest (Using Formula)