ICSE Chemistry Previous Year Question Paper 2014 Solved for Class 10
ICSE Paper 2014
CHEMISTRY
(Two Hours)
Answers to this Paper must be written on the paper provided separately.
You will not be allowed to write during the first 15 minutes.
This time is to be spent in reading the Question Paper.
The time given at the head of this Paper is the time allowed for writing the answers.
Section I is compulsory. Attempt any four questions from Section II.
The intended marks for questions or parts of questions are given in brackets [ ].
SECTION-I (40 Marks)
(Attempt all questions from this Section)
Question 1:
(a) Choose the correct answer from the options given below:
- Ionisation Potential increases over a period from left to right because the:
(A) Atomic radius increases and nuclear charge increases
(B) Atomic radius decreases and nuclear charge decreases
(C) Atomic radius increases and nuclear charge decreases
(D) Atomic radius decreases and nuclear charge increases. - A compound X consists of only molecules. Hence X will have:
(A) A crystalline hard structure
(B) A low melting point and low boiling point
(C) An ionic bond
(D) A strong force of attraction between its molecules. - When fused lead bromide is electrolysed we observe:
(A) a silver grey deposit at anode and a reddish brown deposit at cathode
(B) a silver grey deposit at cathode and a reddish brown deposit at anode
(C) a silver grey deposit at cathode and reddish brown fumes at anode
(D) silver grey fumes at anode and reddish brown fumes at cathode. - The main ore used for the extraction of iron is:
(A) Haematite (B) Calamine
(C) Bauxite (D) Cryolite - Heating an ore in a limited supply of air or in the absence of air at a temperature just below its melting point is known as:
(A) smelting (B) ore dressing
(C) calcination (D) bessemerisation - If an element A belongs to Period 3 and Group II then it will have:
(A) 3 shells and 2 valence electrons
(B) 2 shells and 3 valence electrons
(C) 3 shells and 3 valence electrons
(D) 2 shells and 2 valence electrons - The molecule containing a triple covalent bond is:
(A) ammonia (B) methane
(C) water (D) nitrogen - The electrolyte used for electroplating an article with silver is:
(A) silver nitrate solution (B) silver cyanide solution
(C) sodium argentocyanide solution (D) nickel sulphate solution - Aluminium powder is used in thermite welding because:
(A) it is a strong reducing agent (B) it is a strong oxidising agent
(C) it is corrosion resistant (D) it is a good conductor of heat - The I.U.PA.C. name of acetylene is:
(A) propane (B) propyne
(C) ethene (D) ethyne [10]
(b) Fill in the blanks from the choices given within brackets:
- The basicity of Acetic Acid is ………….. (3, 1, 4)
- The compound formed when ethanol reacts with sodium is …………. (sodium ethanoate, sodium ethoxide, sodium propanoate).
- Quicklime is not used to dry HCl gas because ………….. (CaO is alkaline, CaO is acidic, CaO is neutral).
- Ammonia gas is collected by (an upward displacement, of air, a downward displacement of water, a downward displacement of air)
- Cold, dilute nitric acid reacts with copper to form …………… (Hydrogen, nitrogen dioxide, nitric oxide). [5]
(c) Give one word or phrase for the following:
- The ratio of the mass of a certain volume of gas to the mass of an equal volume of hydrogen under the same conditions of temperature and pressure.
- Formation of ions from molecules.
- Electrolytic deposition of a superior metal on a baser metal.
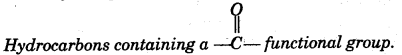
- The amount of energy released when an atom in the gaseous state accepts an electron to form an anion. [5]
(d) Match the options A to E with the statements (i) to (v): [5]
| A | alkynes | (i) | No. of molecules in 22.4 dm3 of carbon dioxide at s.t.p. |
| B | alkane | (ii) | An element with electronic configuration 2, 8, 8, 3 |
| C | iron | (iii) | CnH2n+2 |
| D | 6.023 × 1023 | (iv) | CnH2n-2 |
| E | metal | (v) | The metal that forms two types of ions |
(e) Write balanced equations for the following:
(i) Action of heat on a mixture of copper and concentrated nitric acid.
(ii) Action of warm water on magnesium nitride.
(iii) Action of concentrated sulphuric acid on carbon.
(iv) Action of dilute hydrochloric acid on sodium sulphide.
(v) Preparation of ethane from sodium propionate. [5]
(f) Distinguish between the following pairs of compounds using the test given within brackets:
- Iron (II) sulphate and iron(III) sulphate (using ammonium hydroxide)
- A lead salt and a zinc salt (using excess ammonium hydroxide)
- Sodium nitrate and sodium sulphite (using dilute sulphuric acid)
- Dilute sulphuric acid and dilute hydrochloric acid (using barium chloride solution)
- Ethane and ethene (using alkaline potassium permanganate solution. [5]
(g) (i) Oxygen oxidises ethyne to carbon dioxide and water as shown by the equation:
2C2H2 + 5O2 → 4CO2 + 2H20
What volume of ethyne gas at s.t.p. is required to produce 8.4 dm3 of carbon dioxide at s.t.p.? [H = 1, C = 12, O = 16]
(ii) A compound made up of two elements X and Y has an empirical formula X2Y. If the atomic weight of X is 10 and that of Y is 5 and the compound has a vapour density 25, find its molecular formula. [5]
Answer:
(a)
- (D) Atomic radius decreases and nuclear charge increases.
- (B) A low melting point and low boiling point.
- (C) A silver grey deposit at cathode and reddish brown fumes at anode.
- (A) Haematite
- (C) Calcination
- (A) 3 shells and 2 valence electrons
- (D) Nitrogen
- (C) Sodium argentocyanide solution
- (A) It is a strong reducing agent.
- (D) Ethyne
(b)
- 1
- Sodium ethoxide
- CaO is alkaline
- a downward displacement of air
- nitric oxide
(c)
- Vapour density
- Ionisation
- Electroplating
- Ketone or Carbonyl compound
- Electron affinity
(d)
- A — (iv)
- B — (iii)
- C — (v)
- D — (i)
- E — (ii)
(e)
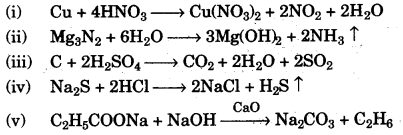
(f)
- Iron II sulphate: Gives dirty green ppt with ammonium hydroxide insoluble in excess.
Iron III sulphate: Gives reddish brown ppt with ammonium hydroxide insoluble in excess. - Lead salt: Gives white ppt with ammonium hydroxide which is insoluble in excess.
Zinc salt: Gives gelatenous white ppt which is soluble in excess ammonium hydroxide. - Sodium nitrate: Colourless vapours of nitric acid which condenses to form nitric acid.
Sodium sulphite: Colourless, gas with smell of burning sulphur, acidic in nature that is sulphur di oxide is released. - With dil. HCl, BaCl2 gives no ppt with dil. H2SO4, BaCl2 gives a white insoluble ppt of BaSO4.
- With ethane, purple colour of potassium permanganate remains unfaded with ethene the purple colour gets decolourised.
(g)

SECTION-II (40 Marks)
(Answer any four questions from this section)
Question 2:
(a) State your observation in each of the following cases:
- When dilute hydrochloric acid is added to sodium carbonate crystals.
- When excess sodium hydroxide is added to calcium nitrate solution.
- At the cathode when acidified aqueous copper sulphate solution is electrolyzed with copper electrodes.
- When calcium hydroxide is heated with ammonium chloride crystals.
- When moist starch iodide paper is introduced into chlorine gas. [5]
(b) Study the figure given below and answer the questions that follow:

- Identify the gas Y.
- What property of gas Y does this experiment demonstrate?
- Name another gas which has the same property and can be demonstrated through this experiment. [3]
(c)
- Name the other ion formed when ammonia dissolves in water.
- Give one test that can be used to detect the presence of the ion produced. [2]
Answer:
(a)
- Brisk effervescence with the release of a colourless odourless gas that extinguish a glowing splint and turns lime water milky i.e., CO2 gas is released.
- A white ppt of Ca(OH)2 is obtained that remains insoluble in excess of NaOH.
- The blue colour of aq.CuSO4 remains unchanged.
- A colourless pungent smelling basic gas i.e., Ammonia is obtained.
- Moist starch iodide paper turns blue black.
(b)
- Hydrogen chloride gas (HCl).
- Y Gas i.e., HCl gas is highly soluble and acidic in nature.
- Ammonia gas.
(c)
- Hydroxyl ion (OH–) other than Ammonium ion.
- Red litmus turns blue/Methyl orange yellow/Phenolphthalein turns pink.
Question 3:
(a) State the conditions required for the following reactions to take place:
- Catalytic hydrogenation of ethyne.
- Preparation of ethyne from ethylene dibromide.
- Catalytic oxidation of ammonia to nitric oxide.
- Any two conditions for the conversion of sulphur dioxide to sulphur trioxide. [5]
(b) State the main components of the following alloys:
- Brass.
- Duralumin.
- Bronze. [3]
(c) Give balanced equations for the following:
(i) Laboratory preparation of nitric acid.
(ii) Preparation of ethanol from monochloroethane and aq. sodium hydroxide. [2]
Answer:
(a)
- In presence of Catalyst like Ni/Pt/Pd etc.
- Heating of ethylene dibromide by using alcoholic KOH.
- In presence of Platinum catalyst at 800 °C.
- In presence of vanadium pentaoxide (V2O5) or Pt as catalyst at 450 °C.
(b)
- Brass: Cu + Zn.
- Duralumin: Al + Cu + Mg + Mn
- Bronze: Cu + Sn.
(c)
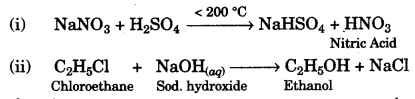
Question 4:
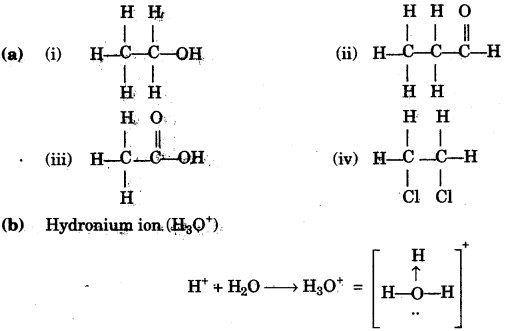
(a) Give the structural formula of the following:
(i) ethanol.
(ii) 1-propanal
(iii) ethanoic acid
(iv) 1, 2, dichloroethane. [4]
(b) Draw the structure of the stable positive ion formed when an acid dissolves in water. [2]
(c) State the inference drawn from the following observations: [4]
- On carrying out the flame test with a salt P a brick red flame was obtained. What is the cation in P?
- A gas Q turns moist had acetate paper silvery black. Identify the gas Q.
- pH of liquid R is 10. What kind of substance is R?
- Salt S is prepared by reacting dilute sulphuric acid with copper oxide. Identify S.
Answer:
(c)
- P → Calcium ion (Ca++)
- Q → Hydrogen sulphide gas (H2S)
- R → Base.
- S → Copper Sulphate (CuSO4).
Question 5:
(a) Name the following:
- The property possessed by metals by which they can be beaten into sheets.
- A compound added to lower the fusion temperature of electrolytic bath in the extraction of aluminium.
- The ore of zinc containing its sulphide. [3]
(b) Give one equation each to show the following properties of sulphuric acid:
(i) Dehydrating property.
(ii) Acidic nature.
(iii) As a non-volatile acid. [3]
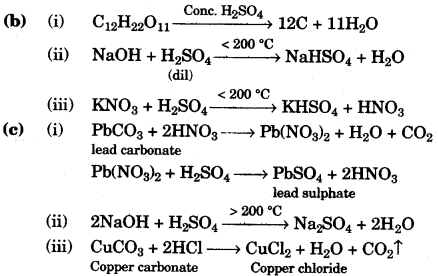
(c) Give balanced chemical equations to prepare the following salts:
(i) Lead sulphate from lead carbonate.
(ii) Sodium sulphate using dilute sulphuric acid.
(iii) Copper chloride using copper carbonate. [3]
Answer:
(a)
- Malleability
- Cryolite (Na3AlF6)
- Zinc Sulphide/Zinc blende (ZnS)
Question 6:
(a) (i) State Avogadro’s law.
(ii) A cylinder contains 68g of ammonia gas at s.t.p.
- What is the volume occupied by this gas?
- How many moles of ammonia are present in the cylinder?
- How many molecules of ammonia are present in the cylinder?
[N-14, H-1] [4]
(b)
- Why do covalent compounds exist as gases, liquids or soft solids?
- Which electrode: anode or cathode is the oxidising electrode? Why? [3]
(c) Name the kind of particles present in:
- Sodium Hydroxide solution.
- Carbonic acid.
- Sugar solution.
Answer:
(a) (i) Under the similar conditions of temperature and pressure, equal volumes of all gases contains equal number of molecules.
(ii) (1) NH3 = 14 + 3 = 17


(b)
- Because the particles/atoms are held by weak Wander Vaal’s forces.
- Anode. Because anode is the oxidising electrode, there is loss of electrons.
(c)
- Ions i.e., Na+ and OH–
- Ions i.e., H+ and CO32–
- Molecules C12H22O11.
Question 7:
(a) An element Z has atomic number 16. Answer the following questions on Z:
- State the period and group to which Z belongs.
- Is Z a metal or a non-metal?
- State the formula between Z and Hydrogen.
- What kind of a compound is this? [5]
(b) M is a metal above hydrogen in the activity series and its oxide has the formula M2O. This oxide when dissolved in water forms the corresponding hydroxide which is a good conductor of electricity. In the above context answer the following:
- What kind of combination exists between M and O?
- How many electrons are there in the outermost shell of M?
- Name the group to which M belongs.
- State the reaction taking place at the cathode.
- Name the product at the anode. [5]
Answer:
(a) Z = 16 = 2, 8, 6.
- Period No. = 3
Group No. = VI A/16 - Non metal.
- Z’s valency = – 2
H = +1 So formula H2Z - Polar Covalent compound.
(b)
- Electrovalent/Ionic compound is formed.
- 1
- M belong to alkali metal group i.e., Group-1.
- M+ + e– → M — Reduction.
- Oxygen gas.