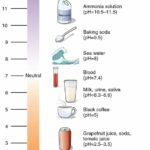
How to calculate concentration of acids and alkalis? Concentrations of Acids and AlkalisA solution is a mixture formed by dissolving a solute in a solvent. Solute + solvent → solution For example, a sugar solution is prepared by dissolving sugar (solute) in water (solvent). By dissolving varying amounts of sugar in a fixed volume of water, sugar solutions of … [Read more...] about How to calculate concentration of acids and alkalis?
Chemistry
Frank ICSE Solutions for Class 10 Chemistry – Alkanes
Frank ICSE Solutions for Class 10 Chemistry - Alkanes PAGE NO : 252 Solution 1:Solution 2:Solution 3:Solution 4:Solution 5:Solution 6:Solution 7:Solution 8:Solution 9:Solution 10:Solution 11:Solution 12:Solution 13:ChemistryBiologyPhysicsMaths … [Read more...] about Frank ICSE Solutions for Class 10 Chemistry – Alkanes
Frank ICSE Solutions for Class 10 Chemistry – Practical Work
Frank ICSE Solutions for Class 10 Chemistry - Practical Work PAGE NO : 282 Solution 1:Solution 2:Solution 3:Solution 4:Solution 5:Solution 7:Solution 8:Solution 9:Solution 10:Solution 1999-1:Solution 2000-1:PAGE NO : 284Solution 2001-1:Solution 2001-2:Solution 2002-1:Solution … [Read more...] about Frank ICSE Solutions for Class 10 Chemistry – Practical Work
Frank ICSE Solutions for Class 10 Chemistry – Carboxylic acid
Frank ICSE Solutions for Class 10 Chemistry - Carboxylic acid PAGE NO : 269 Solution 1:Solution 2:Solution 3:Solution 4:Solution 5:Solution 6:Solution 7:Solution 8:Solution 9:Solution 10:Solution 11:Solution 1999-1:Solution 1999-2:Solution 2000-1:Solution 2000-2:Solution … [Read more...] about Frank ICSE Solutions for Class 10 Chemistry – Carboxylic acid
Frank ICSE Solutions for Class 10 Chemistry – Alcohols
Frank ICSE Solutions for Class 10 Chemistry - Alcohols PAGE NO : 264 Solution 1:Solution 2:Solution 3:Solution 4:Solution 5:Solution 6:Solution 7:Solution 8:ChemistryBiologyPhysicsMaths … [Read more...] about Frank ICSE Solutions for Class 10 Chemistry – Alcohols